Figures & data
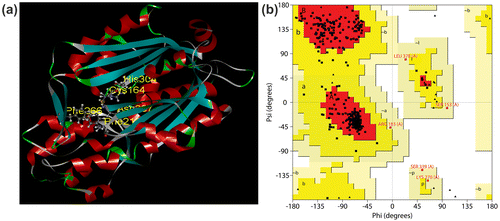
Figure 2. Protein model and stereochemical study of CfCHS. (a) The predicted tertiary structure of CfCHS. The catalytic residues (Cys164, His304, and Asn337) and two highly conserved residues (Phe 216 and Phe 266) are indicated. (b) Ramachandran plot of CfCHS. The plot calculations on the 3D models of C. forskohlii CfCHS proteins were computed with the PROCHECK server. Most favored regions are colored red (A, B, L), additional allowed (a, b, l, p), generously allowed (a, b, l, p), and disallowed regions are indicated as yellow, light yellow, and white regions, respectively.