Figures & data
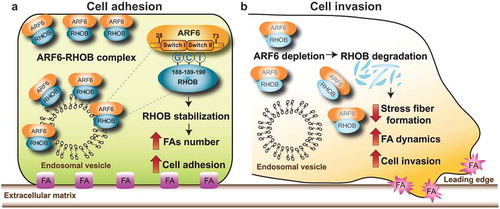
Figure 1. Mechanisms though which ARF6-RHOB regulates breast cancer cell invasion. (a) ARF6 interacts with RHOB. The interaction requires the tripeptide “GCI” (glycine, cysteine, and, isoleucine) residues (188–190) of RHOB and the effector domain regions, Switch I and Switch II (28–73) of ARF6. ARF6 is essential for RHOB protein stability and the regulation of its subcellular localization. Specific targeting of ARF6 to the plasma membrane or endosomal vesicle promotes the recruitment and colocalization of RHOB to these membrane microdomains, thereby controlling cell adhesion by increasing focal adhesion (FA) formation. (b) ARF6 depletion promotes the loss of RHOB from endosomal vesicles and plasma membrane, leading to RHOB degradation. Inhibition of ARF6-RHOB complexes reduces stress fiber formation, increases FA dynamics, promoting breast cancer cell invasion.