Figures & data
Figure 1. Effectiveness with 95% confidence interval (CI) of the BNT16b2 mRNA (Pfizer-BioNTech) vaccine in preventing SARS-CoV-2 infection during period 3 (1–14 February 2021) and 4 (15–28 February 2021).
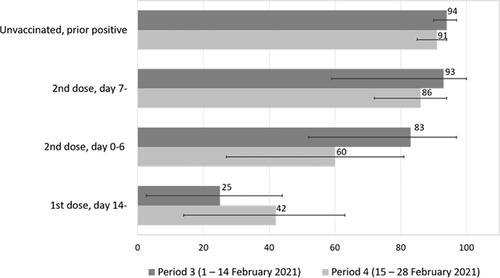
Table 1. Baseline characteristics of the study cohort on 27 December 2020 when COVID-19 vaccination started, stratified by vaccination status on 28 February 2021.
Table 2. Effectiveness of the BNT16b2 mRNA (Pfizer-BioNTech) vaccine in preventing SARS-CoV-2 infection during period 4 (15–28 February 2021).
