Figures & data
Table 1. Distribution of demographics and clinical features between patients and healthy controls.
Table 2. Neurological signs and subjective symptoms during the acute phase, discharge and both follow-ups.
Figure 1. Rankin Scale (a) and Glasgow Outcome Scale (b) during discharge depending on disease severity.
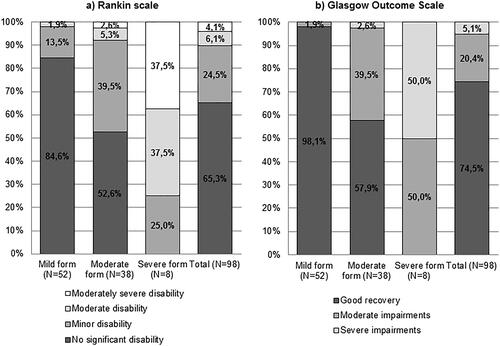
Figure 2. Comparison of different cognitive domains between tick-borne encephalitis (TBE) patients and healthy controls depending on age, 6 months after TBE (*p < 0.05, **p < 0.01).
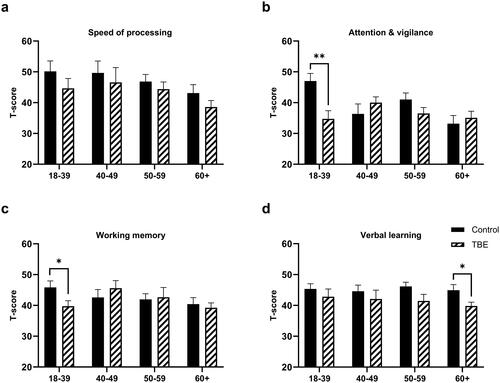
Figure 3. Comparison of different cognitive domains and Overall composite score between tick-borne encephalitis (TBE) patients and healthy controls depending on age, 6 months after TBE (*p < 0.05, **p < 0.01).
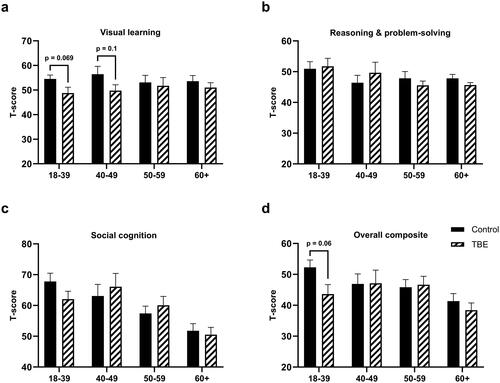
Table 3. Comparison of neurocognitive domains according to the severity of TBE at the time of first (6 months after the acute phase) follow-up controlling for age effect.
Table 4. Changes in TBE patients’ neurocognitive functioning from 1st to 2nd follow-up. Paired sample t-test.
Supplemental Material
Download MS Word (26.5 KB)Supplemental Material
Download MS Word (19.9 KB)Supplemental Material
Download MS Word (66.1 KB)Supplemental Material
Download MS Word (13.9 KB)Supplemental Material
Download MS Word (13.5 KB)Supplemental Material
Download MS Word (13.8 KB)Data availability statement
The participants of this study did not give written consent for their data to be shared publicly, so due to the sensitive nature of the research supporting data is not available.
