Figures & data
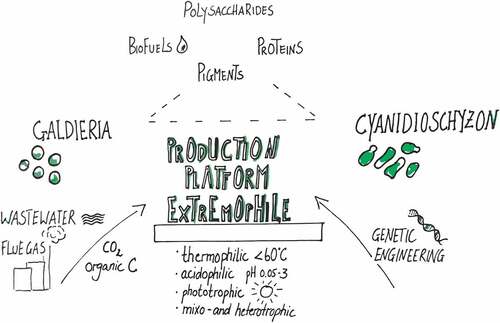
Table 1. Accessibility of Galdieria and Cyanidioschyzon for biotechnological applications.