Figures & data
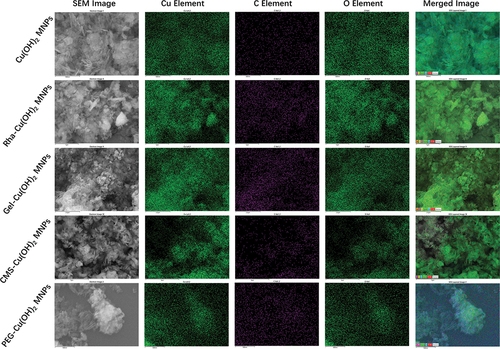
Figure 1. Characterization of copper pesticide NPs. SEM images of Cu(OH)2 NPs (a), Cu(OH)2 NWs (b), CuO NPs (c), rha-coated Cu(OH)2 NPs (d), gel-coated Cu(OH)2 NPs (e), CMS-coated Cu(OH)2 NPs (f), and PEG-coated Cu(OH)2 NPs (g), respectively.

Figure 4. Cu2+ release kinetic profiles of different Cu-based NPs. (a) Cu2+ release profiles of Cu(OH)2 NPs, Cu(OH)2 NWs, and CuO NPs; (b) Cu2+ release profiles of rha, gel, CMS, and PEG-coated Cu(OH)2 NPs.
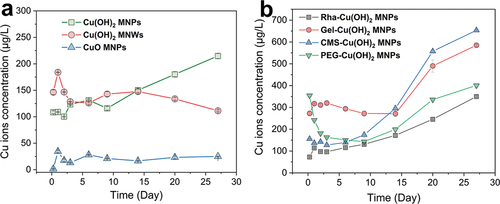
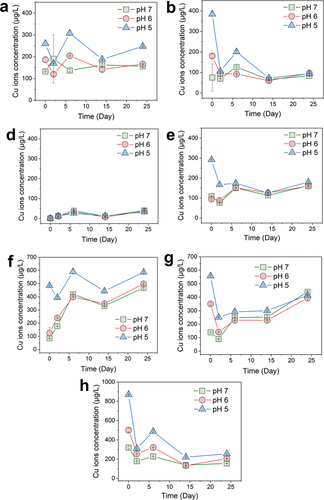
Figure 5. Cu2+ release monitoring of Cu-based NPs at different pH values. Characterization of copper pesticide NPs. Cu2+ release kinetic curves of Cu(OH)2 NPs (a), Cu(OH)2 NWs (b), CuO NPs (c), rha-coated Cu(OH)2 NPs (d), gel-coated Cu(OH)2 NPs (e), CMS-coated Cu(OH)2 NPs (f), and PEG-coated Cu(OH)2 NPs (g), respectively.