Figures & data
Figure 1. Preparation of polyacrylic acid (PAA) nanoparticles through reverse micellar polymerization.
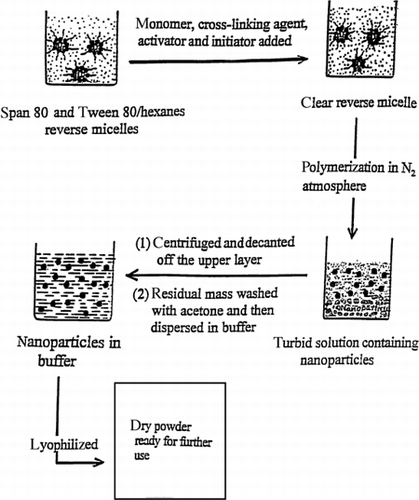
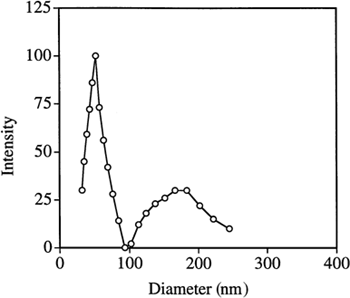
Table 1. Polyacrylic Acid Particle Sizes in Water as Measured by Quasielastic Laser Light Scattering
Figure 4. Quasielastic laser light scattering by PAA nanoparticles in phosphate buffer (50 mM, pH 7.0). Particle sizes are shown before and after lyophilization.
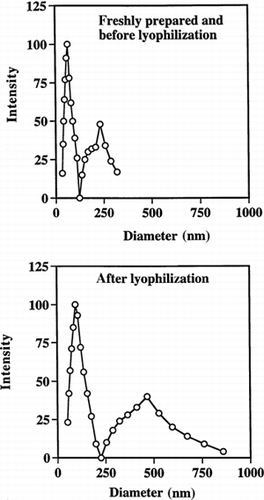
Figure 5. Quasielastic laser light scattering by PAA nanoparticles in phosphate buffer (50 mM, pH 7.0). Particles sizes are shown before and after storage.
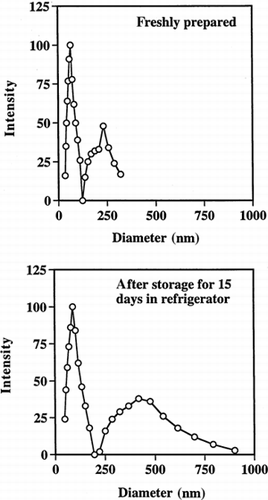
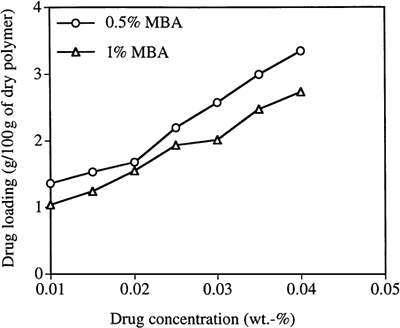
Figure 6. Loading timolol maleate (TM) into PAA nanoparticles as a function of TM concentration in the loading solution. (MBA is the crosslinker.)