Figures & data
Figure 1. Chemical structures, point groups (in parentheses), and triplet excited energies of PAHs with four fused benzene rings.
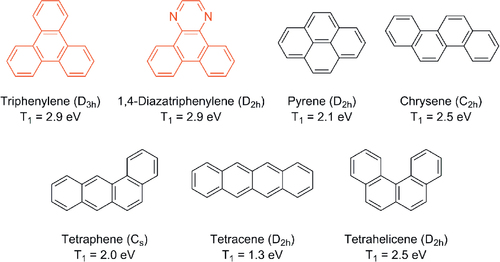
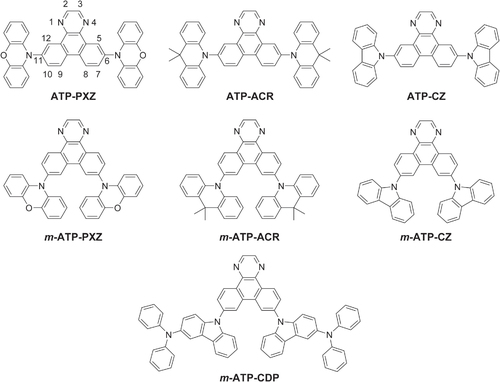
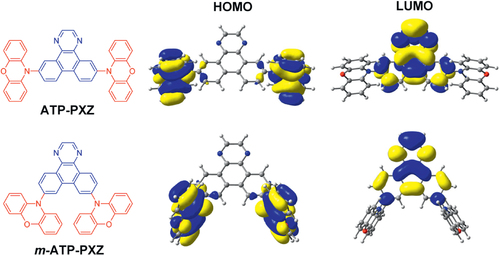
Table 1 Time-dependent density functional theory (TD-DFT) calculation results for ATP-based molecules.
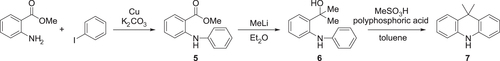
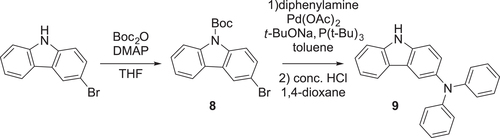
Scheme 3. Synthesis of ATP-based D–A–D-type molecules. Reagents and conditions: (a) N-bromosuccinimide, conc. H2SO4, RT, 2 h (85%); (b) ethylenediamine, EtOH, 80 °C, 1 h, then AcOH, Air, 80 °C, 2 h (56% for 2, 56% for 4); (c) Ar2NH, Pd(OAc)2, P(t-Bu)3, K2CO3, toluene, 24 h, 110 °C; (d) Br2, benzoylperoxide, nitrobenzene, 130 °C, 3 h (85%); (e) Ar2NH (donor units), Pd2(dba)3·CHCl3, N-phenyl-2-(di-t-butylphosphino)indole (cataCXium® PIntB), t-BuONa, toluene, 24 h, 110 °C.
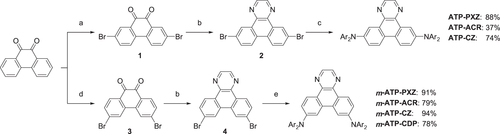
Table 2 Photophysical properties of ATP-based luminescent materials.
Figure 7. Streak image and PL spectra of a 6 wt%-ATP–PXZ:CBP film taken at 300 K showing the prompt (fluorescence, black) and delayed (TADF, red) components. Each dot in the streak image corresponds to the photon count of PL.
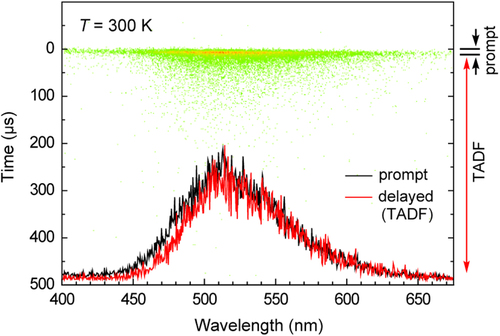
Figure 8. (a) External EL quantum efficiency versus current density curves and (b) current density–voltage–luminance (J–V–L) characteristics of TADF–OLEDs with device configurations of ITO/α-NPD/6 wt%-emitter:host/TPBi/LiF/Al for ATP–PXZ and m-ATP–PXZ, and ITO/α-NPD/mCP/6 wt%-emitter:host/PPT/TPBi/LiF/Al for ATP–ACR and m-ATP–ACR.
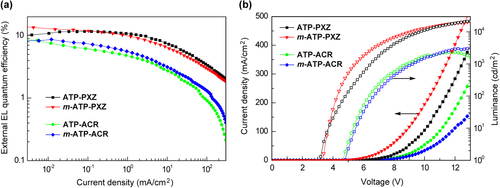
Table 3 Performance of TADF-based OLEDsTable Footnote a.