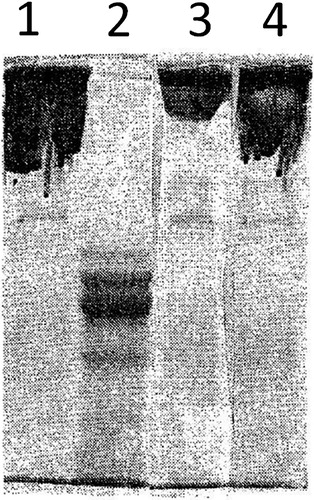Figures & data
Figure 1. Effect of trivalent iron (ferric chloride) and divalent iron (ferrous sulfate) on the generation of hydroxyl radicals measured as fluorescence intensity of hydroxy-derivative of coumarin.
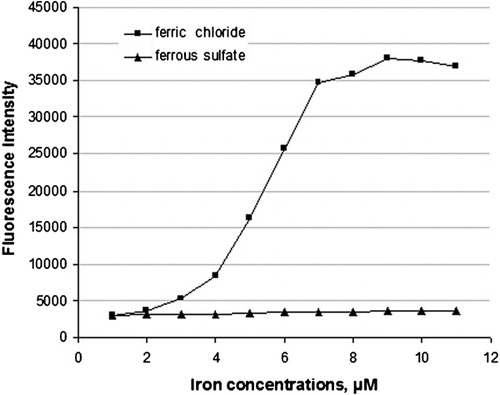
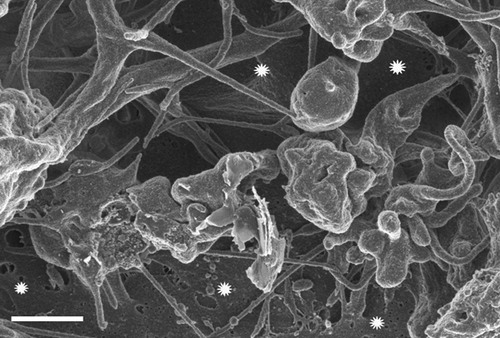
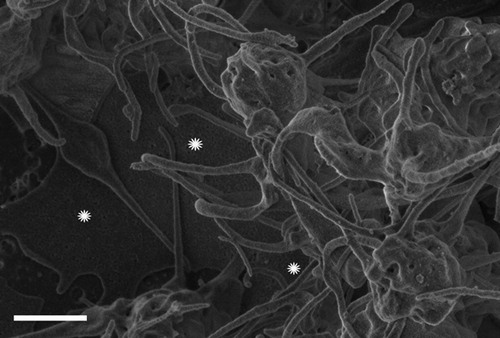
Figure 2. Conversion of soluble fibrinogen into insoluble aggregate, induced with iron and monitored as optical density OD values.
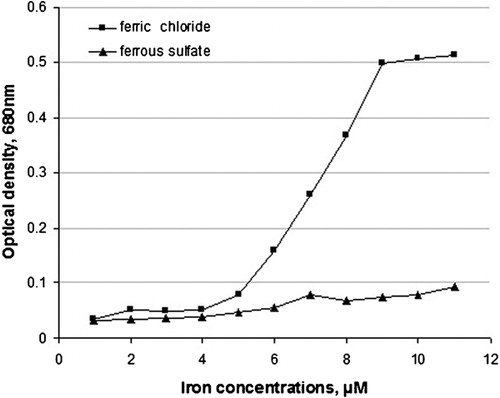
Figure 3. SDS-polyacrylamide gel electrophoresis of native fibrinogen unreduced – line 1; reduced – line 2; and of iron-modified fibrinogen unreduced – line 3; reduced – line 4).