Figures & data
Figure 1. Preferred reporting items for systematic reviews and meta-analysis flow diagram – study selection and exclusion.
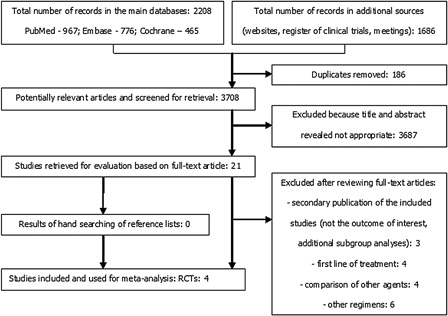
Table 1. Characteristics of the included randomized controlled trials
Table 2. Summary of the efficacy and safety outcomes of the included studies
Figure 2. Meta-analysis of progression-free survival (PFS) for targeted agents used as monotherapy or combined therapy in patients with relapsed or refractory MM.
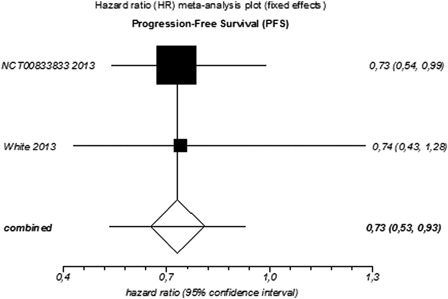
Figure 3. Meta-analysis of overall survival (OS) for targeted agents used as monotherapy or combined therapy in patients with relapsed or refractory MM.
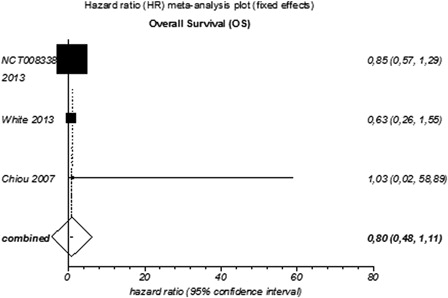
Figure 4. Meta-analysis of any adverse events (AEs) for targeted agents used as monotherapy or combined therapy in patients with relapsed or refractory MM.
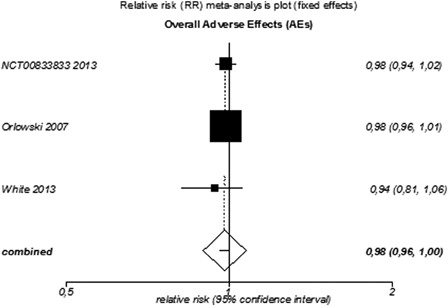
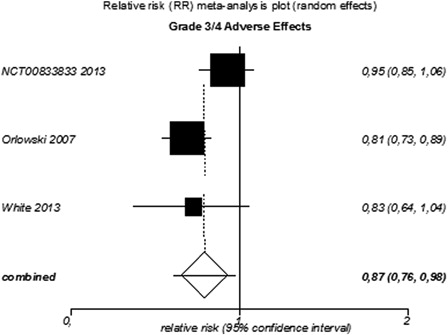
Figure 5. Meta-analysis of grade 3/4 AEs for targeted agents used as monotherapy or combined therapy in patients with relapsed or refractory MM.