Figures & data
Table 1 Demographics of the participants (n = 346)
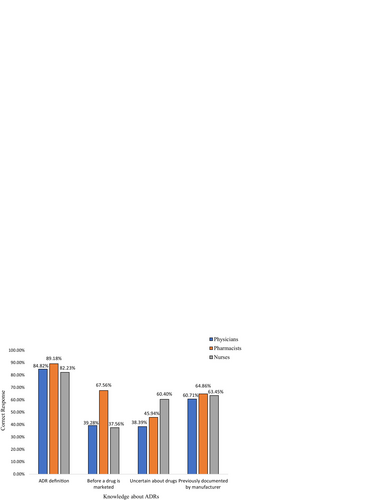
Table 2 Knowledge of HCPs about adverse drug reaction reporting
Table 3 Types of ADRs to be reported
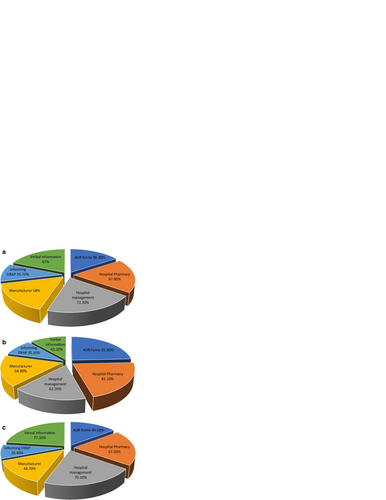
Fig. 2 a Types of ADRs to be reported (by physicians). b Types of ADRs to be reported (by pharmacists). c Types of ADRs to be reported (by nurses)
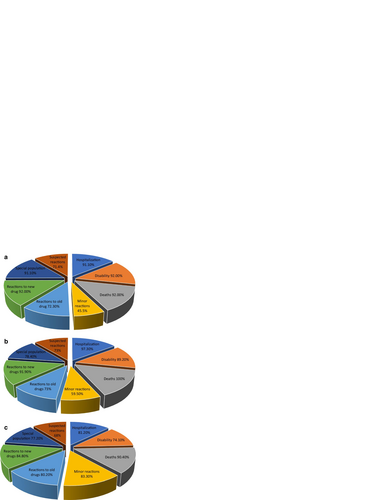
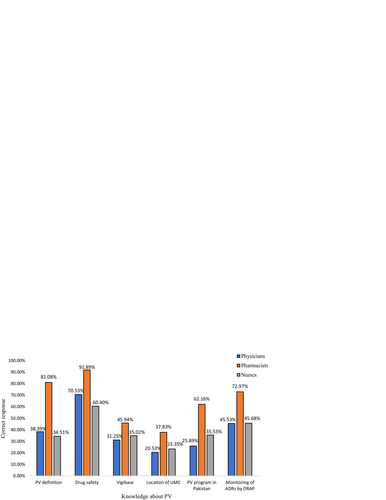
Table 4 Knowledge about pharmacovigilance
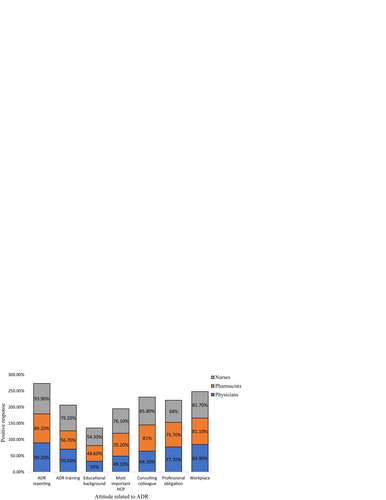
Table 5 Attitudes related to ADR reporting
Table 6 Practices related to ADR reporting
Fig. 5 a Practices of ADR reporting (by physicians). b Practices of ADR reporting (by pharmacists). c Practices of ADR reporting (by nurses)
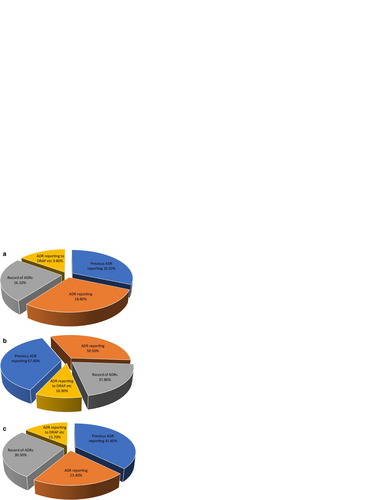
Table 7 Practices related to the modes of ADR reporting
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.