Figures & data
Table 1 Biosimilars available in ambulatory care in Belgium (May 2021) [Citation10]
Table 2 Community pharmacists: participants’ characteristics
Fig. 1 Self-assessed competence of community pharmacists to dispense biologicals (in general) and biosimilars (in particular). Anti-TNF anti-tumor necrosis factor, N number. Statistical testing: ‡: when testing for differences in self-assessed competence in dispensing biological medicines in general versus biosimilars in particular, a statistical difference was found for statement 2 (Additional file 2: Table S4). †: when testing for differences in self-assessed competences in dispensing biological medicines between more recently graduated and more senior community pharmacists (more (N = 47) versus less than 20 years (N = 78) of pharmacy experience), a statistical significant difference was found for statement 1 (Additional file 2: Table S5). When testing for differences in self-assessed competences in dispensing biosimilars between more recently graduated and more senior community pharmacists (more (N = 17) versus less than 20 years (N = 19) of pharmacy experience), no statistical significant difference were found for any of the statements (Additional file 2: Table S6)
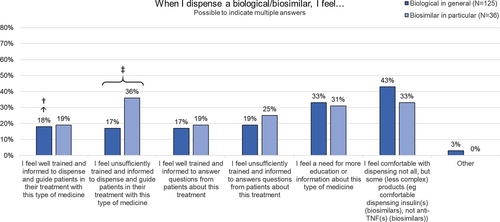
Fig. 2 Perceived challenges when dispensing/prescribing a biological (in general) or a biosimilar (in particular) among community pharmacists and physicians. N number, Q questions, RP reference product
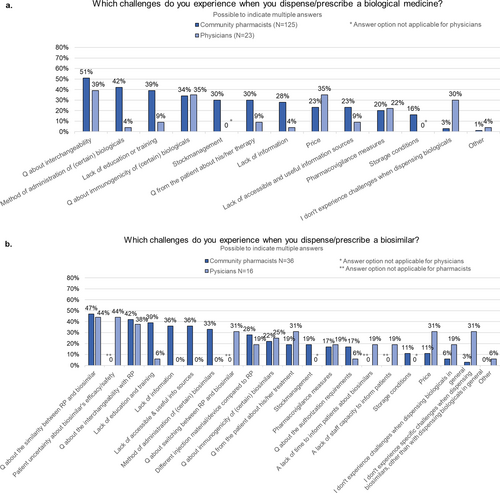
Fig. 3 Community pharmacist and physician views on interchangeability. Interchangeability: interchangeability refers to the possibility of exchanging one medicine for another medicine that is expected to have the same clinical effect. This could mean replacing a reference product with a biosimilar (or vice versa) or replacing one biosimilar with another. N number, RP reference product
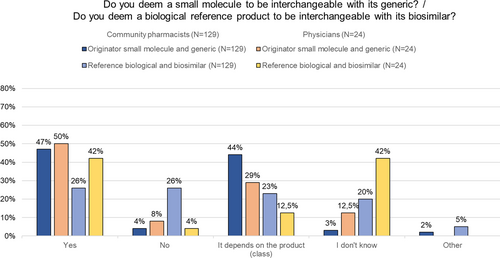
Fig. 4 Questions about (automatic) substitution to community pharmacists and physicians. Automatic substitution: the pharmacist dispenses one medicine instead of another equivalent and interchangeable medicine at pharmacy level without consulting the prescriber. N number, RP reference product
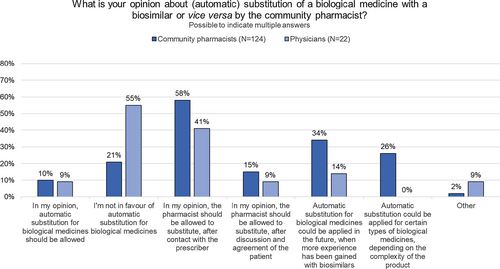
Fig. 5 Information need about biologicals in general and biosimilars in specific among community pharmacists. EMA European Medicines Agency, FAMHP Federal Agency for Medicines and Health Products (Belgian National Competent Authority), KOL key opinion leader, LMWH low molecular weight heparins, N number, NIHDI National Institute for Health and Disability Insurance (Belgian national health insurer), TNF-alfa blockers tumor necrosis factor-alfa blockers
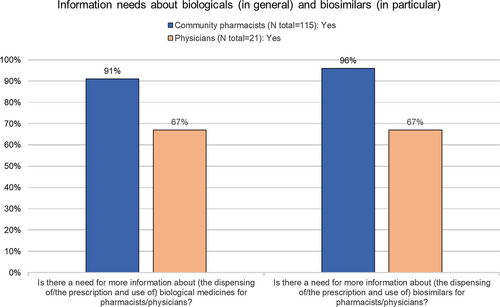
Table 3 Physicians: participants’ characteristics
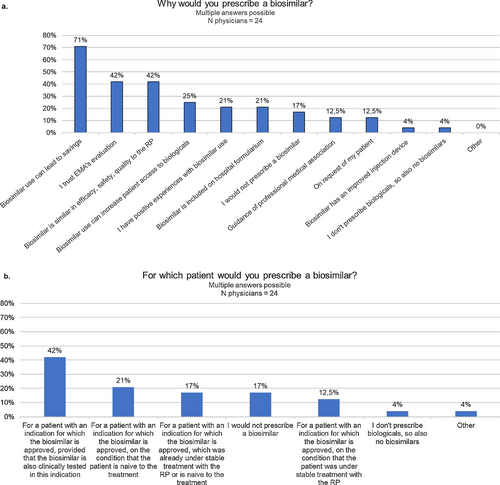
Fig. 6 Questions about the motivation of physicians to prescribe biosimilars. EMA European Medicines Agency, N number, RP reference product
Additional file 1: Figure S1.
Download MS Word (1.5 MB)Additional file 2: Table S1.
Download MS Word (33 KB)Additional file 3:
Download MS Word (27.9 KB)Availability of data and materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
