Figures & data
Table 1.
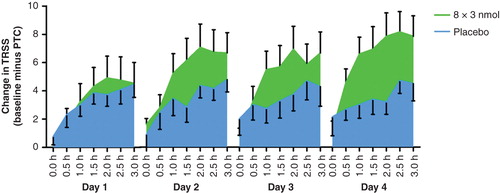
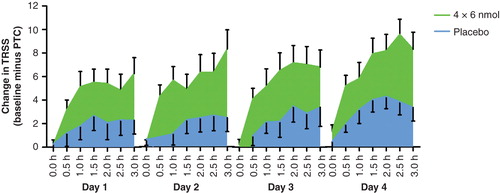
Summary of clinical studies conducted with cat allergen.
Table 2.
Four Phase II prospective, randomized, double-blind, placebo-controlled studies are summarized.