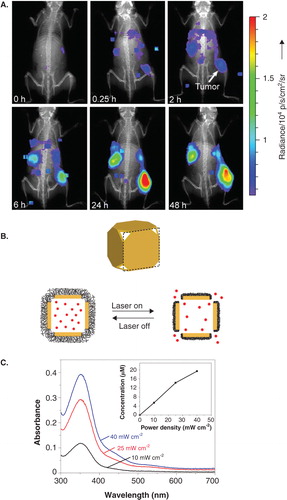
Figures & data
Figure 1. A. Schematic of a device and cross section of the membrane portion. B. Sustained release of aspart from a typical device with triggering by applying heat or by laser irradiation. C. Pulsatile release from the same type of device using 30-min laser pulses (continuous wave, 808 nm, 570 mW/cm2). D. Effect of irradiation on blood glucose levels in diabetic Sprague–Dawley rats.
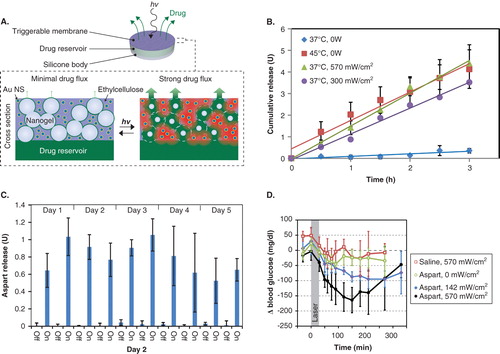
Figure 2. A. Luminescence imaging of whole mice showing accumulation of PEG-coated radioluminescent 198Au nanoshells at tumor sites. B. Schematic of gold nanocage controlled-release system. Upon NIR irradiation, the pNIPAm shell (black) collapses so that the drug (red) is released. C. Release of model drug after triggering for 2 min at various irradiances.