Figures & data
Figure 2. The risk management process for addressing use-related risks with the redesigned follitropin alfa pen.
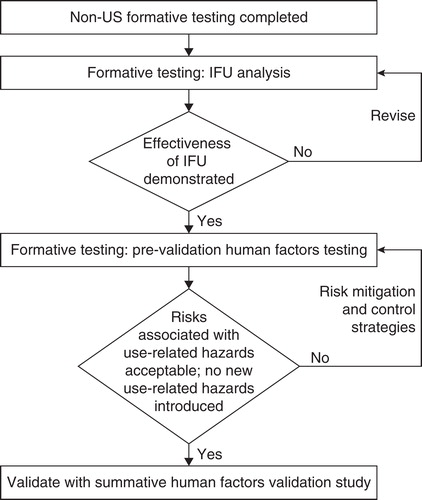
Figure 3. Modification of the redesigned pen device. Change from conical (A) to cylindrical (B) needle cap to mitigate use error observed in the pre-validation studies.

Table 1. Demographic characteristics and prior injection experience of participants in the validation studies.
Table 2. Critical use errors in A) validation study and B) mitigation validation study.