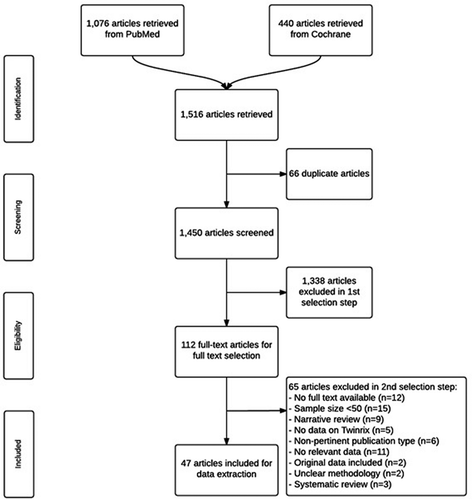
Figures & data
Table 1. Three presentations of combined vaccine against hepatitis A and B.
Table 2. Immunogenicity of TwinrixTM in adults.
Table 3. Immunogenicity of TwinrixTM in children.
Table 4. Immunogenicity of TwinrixTM following an alternative schedule.
Table 5. Immunogenicity of Twinrix PediatricTM in children and adolescents.
Table 6. Immunogenicity of Twinrix PediatricTM in children following an alternative schedule.
Table 7. Immunogenicity of AmbirixTM in children and adolescents.
Table 8. Immunogenicity of AmbirixTM in adolescents following an alternative schedule.
Table 9. Comparison between TwinrixTM and concomitant administration of monovalent hepatitis A and B vaccines.
Table 10. Comparison between Twinrix pediatricTM and concomitant administration of monovalent hepatitis A and B vaccines.
Table 11. Comparison between AmbirixTM and concomitant administration of monovalent hepatitis A and B vaccines.
Table 12. Solicited and unsolicited adverse events following immunization with TwinrixTM, Twinrix pediatricTM, or AmbirixTM.
Table A1. Study characteristics.