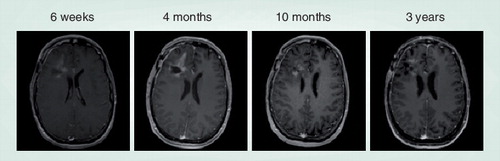
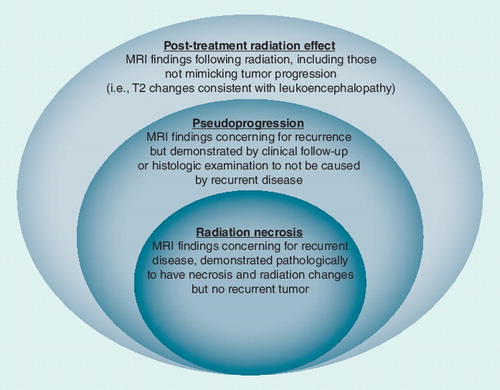
Figures & data
Table 1. Rates of pseudoprogression in studies of radiotherapy plus temozolomide.
Table 2. Median survival (in months) in patients with high-grade glioma undergoing adjuvant radiotherapy with concurrent temozolomide, as assessed by early radiographic findings and subsequent determination of pseudoprogression.
Table 3. Overview of imaging techniques utilized for discriminating between early progressive disease and pseudoprogression.
Activity Evaluation: Where 1 is strongly disagree and 5 is strongly agree