Figures & data
IPD: Invasive pneumococcal disease; PCV: Pneumococcal conjugate vaccine; PPV: Pneumococcal polysaccharide vaccine.
Data taken from Citation[11,25].
![Figure 2. Observed and modeled seroepidemiological changes in the incidence of pneumococcal serotypes causing invasive pneumococcal disease over time from 2005 to 2021 in Germany.IPD: Invasive pneumococcal disease; PCV: Pneumococcal conjugate vaccine; PPV: Pneumococcal polysaccharide vaccine.Data taken from Citation[11,25].](/cms/asset/3c59fe36-3cd8-45d8-af52-290f484558ab/ierp_a_11215585_f0002_b.jpg)
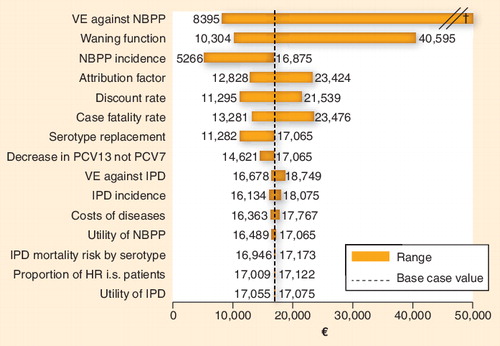
Attribution factor refers to the proportion of nonbacteremic pneumonia having pneumococcal origin.
†The maximum value obtained in the sensitivity analysis was €118,382 per quality-adjusted life-year gained, when vaccine effectiveness of 23-valent pneumococcal polysaccharide vaccine against NBPP was assumed to be 0%. The incremental cost–effectiveness ratio was €75,664, €41,789 and €22,848 per quality-adjusted life-year gained, when vaccine effectiveness of 23-valent pneumococcal polysaccharide vaccine against NBPP was assumed to be 5, 15 and 30% in the elderly and at-risk immunocompetent adults, respectively.
HR: Hazard ratio; IPD: Invasive pneumococcal disease; i.s.: Immunosuppressed; NBPP: Nonbacteremic pneumococcal pneumonia; PCV: Pneumococcal conjugate vaccine; VE: Vaccine effectiveness.
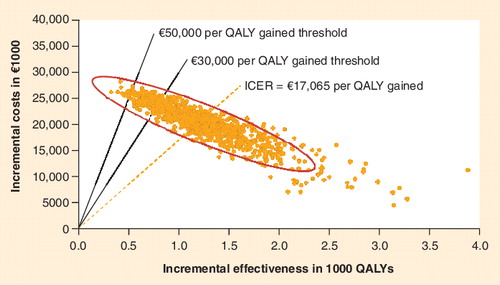
ICER: Incremental cost–effectiveness ratio; QALY: Quality-adjusted life year.
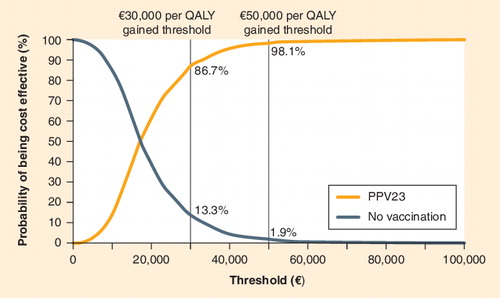
PPV23: 23-valent pneumococcal polysaccharide vaccine; QALY; Quality-adjusted life-year.