Figures & data
Figure 1 Structure and Mechanism of Action of Antibody-Drug Conjugates in the Treatment of Breast Cancer.
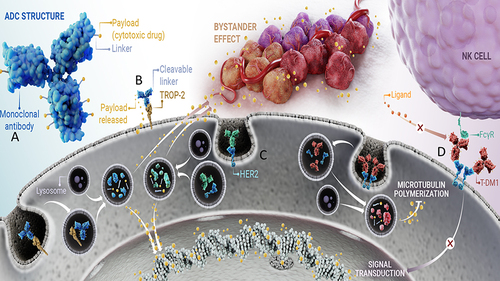
Table 1 Pivotal Trials with ADCs for Breast Cancer Treatment
Table 2 Ongoing Trials: HER2-Positive Breast Cancer
Table 3 Ongoing Trials: Triple Negative Breast Cancer
Table 4 Ongoing Trials: Hormone Receptor Positive Breast Cancer
