Figures & data
Table 1 Monoclonal antibodies in various stages of clinical investigation for the treatment of multiple myeloma
Figure 1 CS1 expression quantified by gene expression (A), and immunohistochemistry (B).
Abbreviations: mAb, monoclonal antibody; MGUS, monoclonal gammopathy of undetermined significance; MM BS, multiple myeloma treated on Total Therapy 2 and 3 protocols at baseline; MM REL, multiple myeloma treated on Total Therapy 2 and 3 protocols at relapse; MM, multiple myeloma; MMCL, myeloma cell lines; NK, natural killer; NPC, normal plasma cells from healthy donors; NT, normal healthy tissue; SMM, smoldering multiple myeloma.
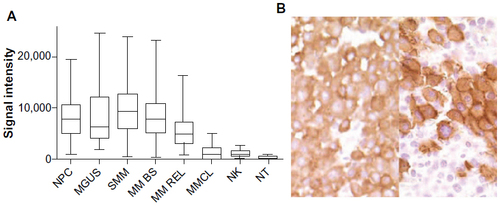
Figure 2 Mechanisms of action of elotuzumab. The primary mechanism of action of elotuzumab against myeloma cells is NK cell-mediated ADCC. Elotuzumab can also interfere with the adhesion of myeloma cells to BMSC, or can induce NK cell activation directly through binding CS1 expressed on NK cells.
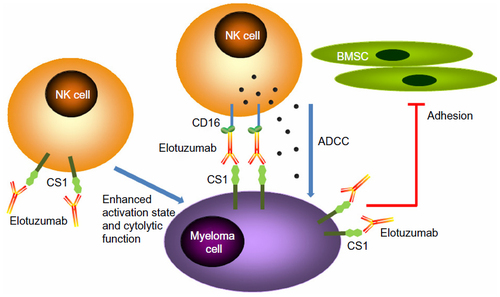
Figure 3 Elotuzumab enhanced both ex vivo expanded NK cell-mediated, and to a lesser extent, resting NK cell-mediated killing of autologous myeloma cells in a standard 4-hour 51Cr release assay.
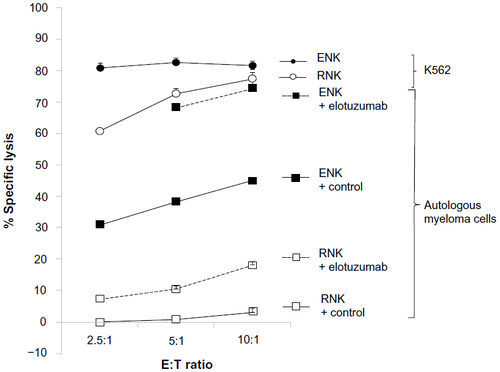
Table 2 Ongoing clinical trials using elotuzumab for the treatment of multiple myeloma
