Figures & data
Figure 1 Decision tree model.
Abbreviations: AE, adverse event; ANI, anidulafungin; pAEV, probability of AEs occurring with VOR monotherapy; pAEVA, probability of AEs occurring with VOR+ANI combination therapy; pMV, probability of mortality with VOR monotherapy; pMVA, probability of mortality with VOR+ANI combination therapy; pSAEV, probability of serious AEs occurring with VOR monotherapy; pSAEVA, probability of serious AEs occurring with VOR+ANI combination therapy; VOR, voriconazole.
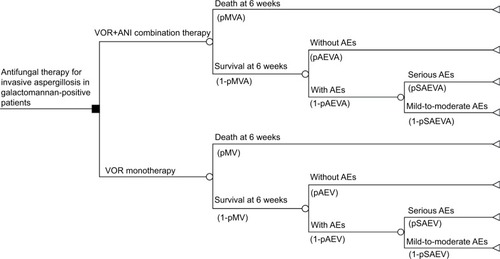
Table 1 Economic model assumptions
Table 2 Drug acquisition cost calculation
Table 3 Deterministic analysis results: base-case
Figure 2 Tornado plot (deterministic sensitivity analysis).
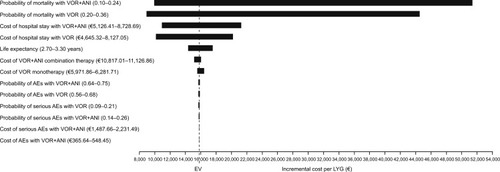
Figure 3 Cost-effectiveness plane (Monte Carlo simulation) at: (A) a willingness-to-pay threshold of €30,000; and (B) a willingness-to-pay threshold of €45,000.
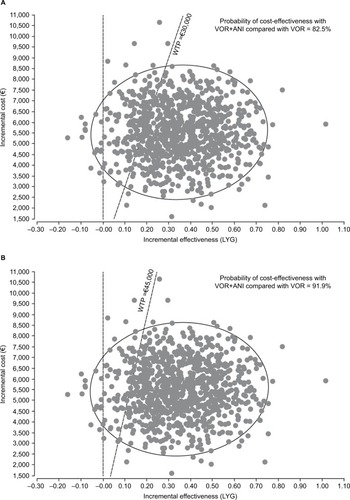
Figure 4 Cost-effectiveness acceptability curve.
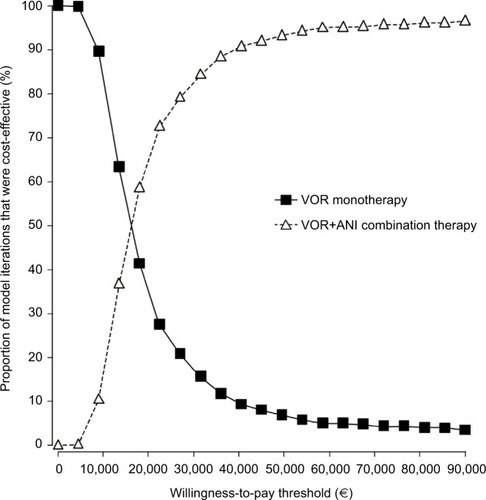
Table 4 Probabilistic analysis results
