Figures & data
Figure 1 Undiscounted (A) and discounted (B) price of the two drug comparators over the model’s time horizon when the comparator drug is already a generic drug.
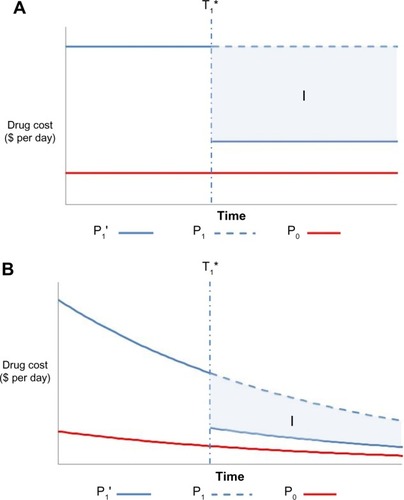
Figure 2 Undiscounted (A) and discounted (B) price of the two drug comparators over the model’s time horizon when both drugs are patented molecules.
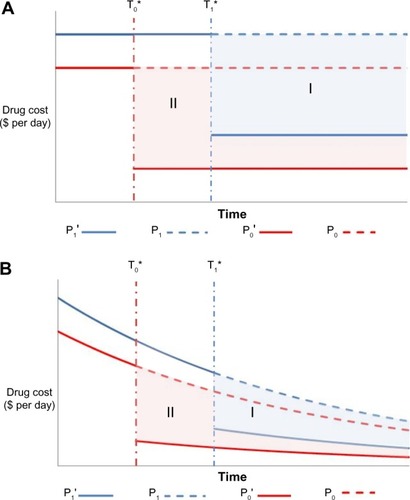
Table 1 Incremental cost of dabigatran etexilate compared to generic warfarin when ignoring and when considering the future introduction of generic dabigatran etexilate
Table 2 Overestimation of the incremental cost based on different times to introduction of generic versions
Table S1 Base case parameters for the case study
