Figures & data
Figure 1 Flow chart of participants.
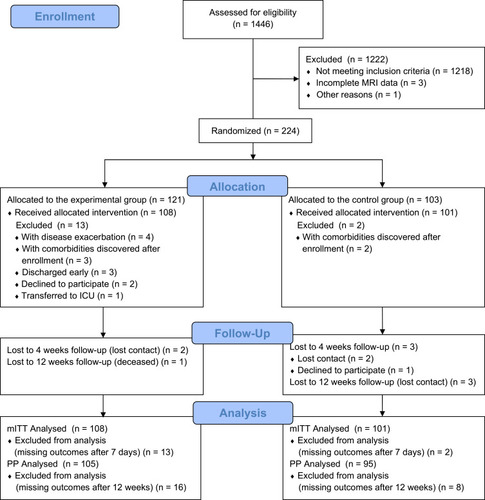
Table 1 Baseline Demographic and Clinical Characteristics Between Groups
Table 2 Between-Group Difference on Primary Outcomes After 7 Days Treatment (PP Analysis, n=200)
Figure 2 Treatment difference and non-inferiority margin. (A) Non-inferiority test for the modified intention-to-treat dataset, n = 209. (B) Non-inferiority test for the per-protocol dataset, n = 200.
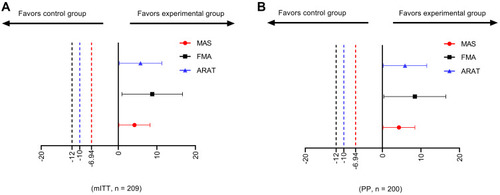
Table 3 Median (IQR) Secondary Outcomes Scores for Each Group, and Results of GEE Analysis (PP Analysis, n = 200; Experimental Group n = 105, Control Group n = 95)
