Figures & data
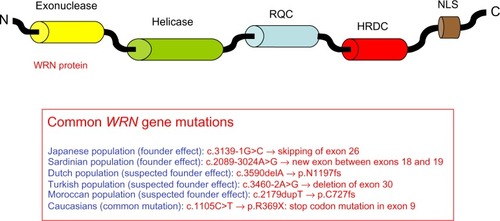
Figure 1 Representation of prelamin A processing and summary of LMNA and ZMPSTE24 mutations leading to classical and atypical progeria.
Abbreviations: HGPS, Hutchinson–Gilford Progeria Syndrome; ZMPSTE24, zinc metalloprotease related to Ste24p; FTASE, farnesyltransferase; RCE1, Ras-converting enzyme 1; ICMT, isoprenylcysteine carboxyl methyltransferase; WS, Werner syndrome.
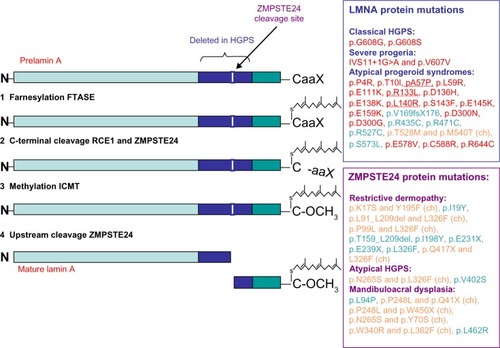
Figure 2 Representation of the WRN protein and summary of the most common WRN mutations leading to Werner syndrome.
Abbreviations: RQC, RecQ C-terminal domain; HRDC, helicase and RNase D C-terminal domain; NLS, nuclear localization signal; N, amino terminal; C, carboxyl terminal.