Figures & data
Table 1 RCTs reporting incidence of recurrent colorectal adenomas
Table 2 Studies reporting posttreatment effect of celecoxib on incidence of recurrent colorectal adenomas after 2 years of off-treatment
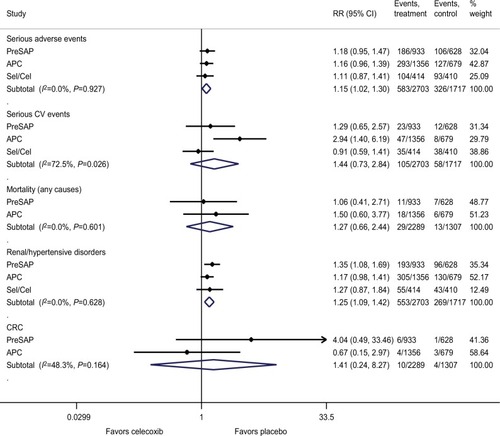
Figure 1 Effects on the primary efficacy outcomes.
Notes: Efficacy outcomes measured cumulatively from baseline, on postrandomization colonoscopy performed within 1 year of discontinuing intervention. Celecoxib at any dose: 400–800 mg/day.
Abbreviations: APC, Adenoma Prevention with Celecoxib trial; PreSAP, Prevention of Colorectal Sporadic Adenomatous Polyps study; Sel/Cel, Selenium and Celecoxib (Sel/Cel) Trial.
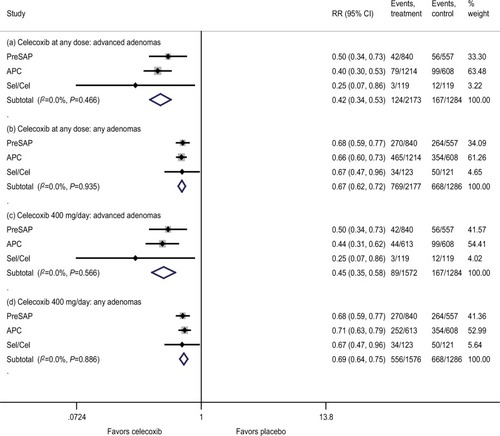
Figure 2 Effects of celecoxib at any dose on the safety outcomes.
Abbreviations: APC, Adenoma Prevention with Celecoxib trial; CRC, colorectal cancer; CV, cardiovascular; PreSAP, Prevention of Colorectal Sporadic Adenomatous Polyps Study; Sel/Cel, Selenium and Celecoxib (Sel/Cel) Trial.