Figures & data
Table 1 Clinicopathologic characteristics of the study subjects
Table 2 iPFS and OS according to subgroups
Figure 1 Kaplan–Meier curves for iPFS in advanced EGFR-mutant NSCLC patients.
Notes: The median iPFS is significantly longer in patients receiving concurrent EGFR-TKIs and WBRT than in those given EGFR-TKIs alone (17.7 vs 11.0 months, P=0.015).
Abbreviations: iPFS, intracranial progression-free survival; NSCLC, non-small-cell lung cancer; TKI, tyrosine kinase inhibitor; WBRT, whole-brain radiotherapy.
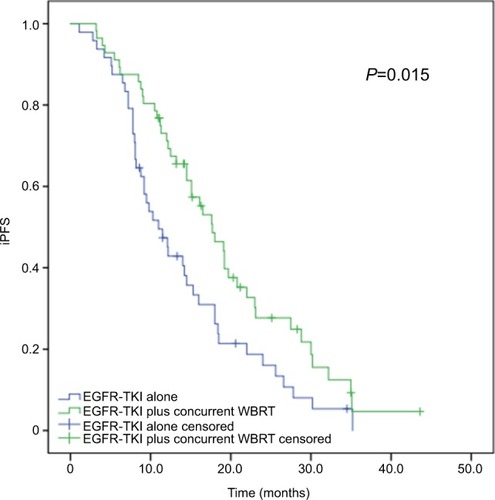
Figure 2 Kaplan–Meier curves for iPFS in advanced EGFR-mutant NSCLC patients with various number of brain metastases.
Note: Patients with more than three brain metastases had significantly shorter median iPFS than those with three or less brain metastases (12.5 vs 18.0 months, P=0.044).
Abbreviations: iPFS, intracranial progression-free survival; NSCLC, non-small-cell lung cancer.
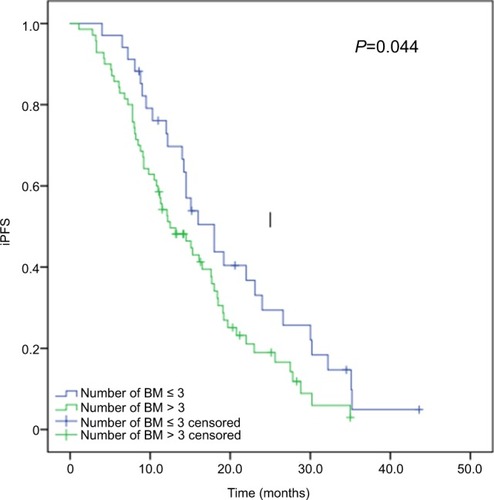
Table 3 Response to therapy in the two study cohorts
Table 4 Univariable and multivariable analyses of covariables associated with intracranial progression-free survival
Figure 3 Kaplan–Meier curves for OS in advanced EGFR-mutant NSCLC patients receiving various treatment regimens.
Note: The median OS did not significantly differ in patients receiving concurrent EGFR-TKIs and WBRT (median OS, 28.1 months; 95% CI, 23.975–32.225 months) and EGFR-TKIs alone (median OS, 24.0 months; 95% CI, 17.428–30.572 months), with the P-value being 0.756.
Abbreviations: NSCLC, non-small-cell lung cancer; OS, overall survival; TKI, tyrosine kinase inhibitor; WBRT, whole-brain radiotherapy.
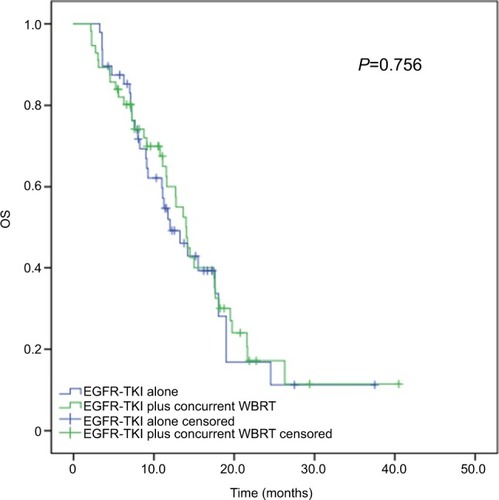
Figure 4 Subgroup analysis of iPFS in advanced EGFR-mutant NSCLC patients with more than three brain metastases.
Note: The median iPFS was significantly longer in patients receiving concurrent EGFR-TKIs and WBRT than in those given EGFR-TKIs alone (17.6 vs 9.2 months, P=0.001).
Abbreviations: iPFS, intracranial progression-free survival; NSCLC, non-small-cell lung cancer; TKI, tyrosine kinase inhibitor; WBRT, whole-brain radiotherapy.
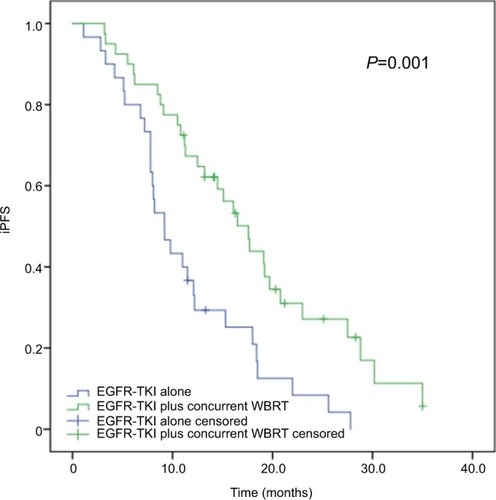
Figure 5 Subgroup analysis of iPFS in advanced EGFR-mutant NSCLC patients with three or less brain metastases.
Note: The median iPFS was comparable between the patients receiving concurrent EGFR-TKIs and WBRT and EGFR-TKIs alone (19.2 vs 14.5 months, P=0.526).
Abbreviations: iPFS, intracranial progression-free survival; NSCLC, non-small-cell lung cancer; TKI, tyrosine kinase inhibitor; WBRT, whole-brain radiotherapy.
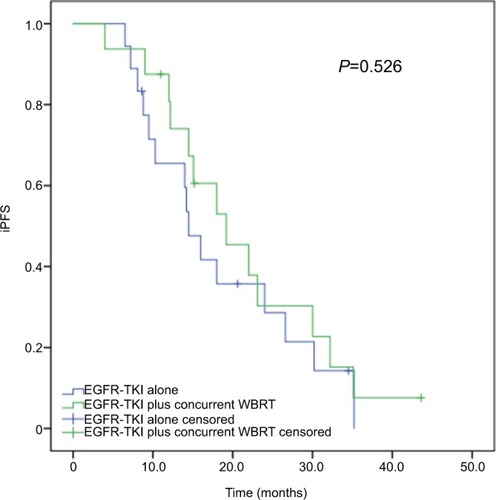
Table 5 Treatment-related toxicities
