Figures & data
Table S1 Baseline characteristics of enrolled studies
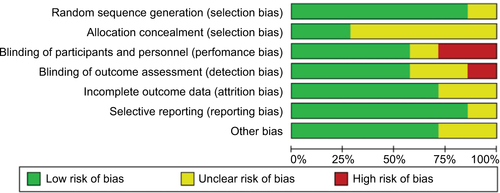
Table S2 Risk of bias in enrolled studies
Figure 2 Frequency of all grade toxicity, including hand-foot syndrome (A), hypocalcemia (B), rash (C), voice change (D), hypertension (E), nausea (F), elevated ALT (G), elevated AST (H), and vomiting (I), among sorafenib and lenvatinib.
Note: ****P<0.0001 indicates statistical significance.
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
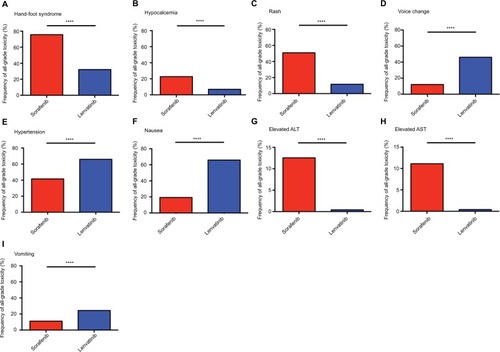
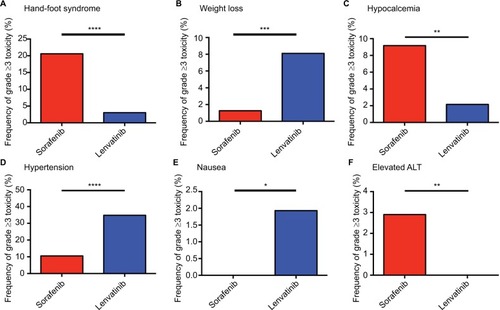
Figure 3 Frequency of grade ≥3 AEs, including hand-foot syndrome (A), weight loss (B), hypocalcemia (C), hypertension (D), nausea (E), and elevated ALT (F), among sorafenib and lenvatinib.
Note: *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 indicate statistical significance.
Abbreviations: AEs, adverse effects; ALT, alanine aminotransferase.