Figures & data
Table 1 Emesis risk level of antineoplastic drugs
Table 2 Pharmacokinetics parameters of oral and transdermal granisetronCitation49,Citation50,Citation61
Figure 1 Plasma concentration of granisetron after application of the transdermal patch of 52 cm2 for 6 days of treatment. Based on study 392MD/11/C.Citation49
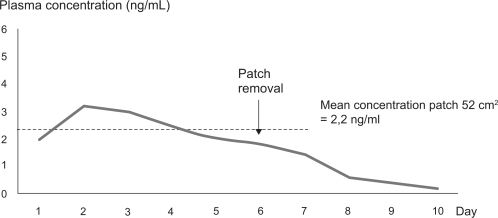
Table 3 Rate of acute emesis control and related-treatment side adverse effects in controlled clinical trialsCitation56,Citation59,Citation60