Figures & data
Figure 1 Study design.
Abbreviations: bid, twice daily; h, hours; IND/GLY, indacaterol/glycopyrronium; od, once daily; SFC, salmeterol/fluticasone.
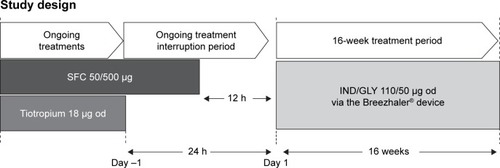
Table 1 Baseline demographics and clinical characteristics
Figure 3 Change from baseline in trough FEV1 at Week 16 (ITT population).
Abbreviations: IND/GLY, indacaterol/glycopyrronium; ITT, intention-to-treat; SFC, salmeterol/fluticasone; Tio, tiotropium.
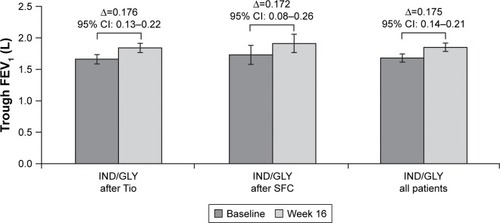
Figure 4 Change from baseline in trough FEV1 at Week 4 (ITT population).
Abbreviations: IND/GLY, indacaterol/glycopyrronium; ITT, intention-to-treat; SFC, salmeterol/fluticasone; Tio, tiotropium.
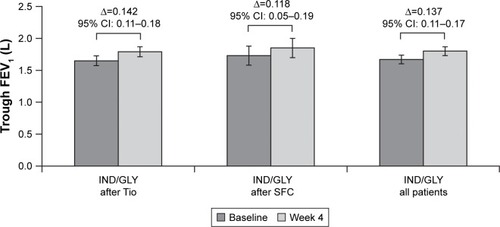
Figure 5 Change from baseline in TDI score at Week 16 (ITT population).
Abbreviations: IND/GLY, indacaterol/glycopyrronium; ITT, intention-to-treat; MCID, minimal clinically important difference; SFC, salmeterol/fluticasone; TDI, transition dyspnea index; Tio, tiotropium.
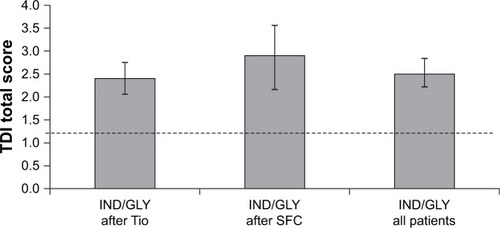
Figure 6 Change from baseline in CAT score at Week 16 (ITT population).
Abbreviations: CAT, COPD assessment test; IND/GLY, indacaterol/glycopyrronium; ITT, intention-to-treat; SFC, salmeterol/fluticasone; Tio, tiotropium.
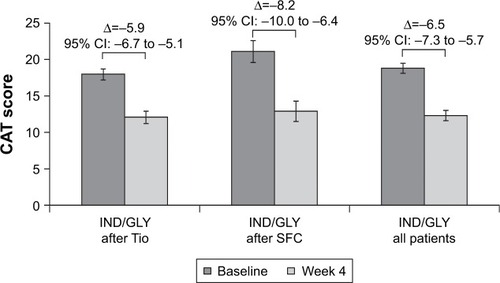
Figure 7 Change from baseline in trough FEV1 at Week 16 by subgroups.
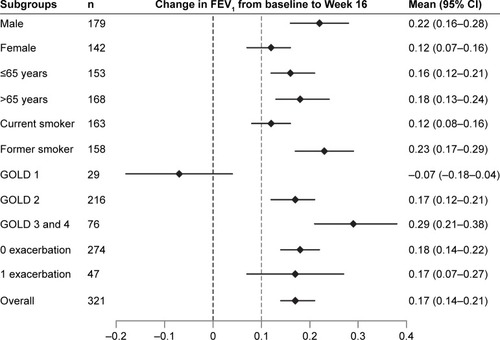
Table 2 Treatment-emergent AEs and SAEs during the study period (safety set)