Figures & data
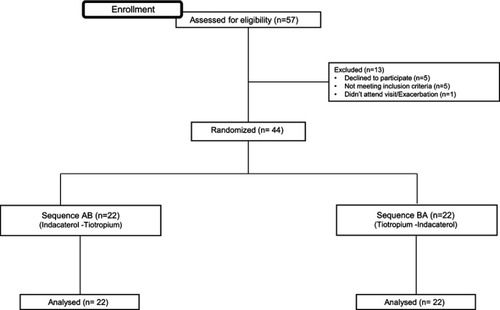
Figure 1 Study design and patient´s allocation of the 2-period cross-over sequences. During both visit 1 and visit 2, the pulmonary function tests measurements were done before and 30, 60, 240 mins, 23:10 and 23:45 hrs after a single-dose of the corresponding long-acting bronchodilator.
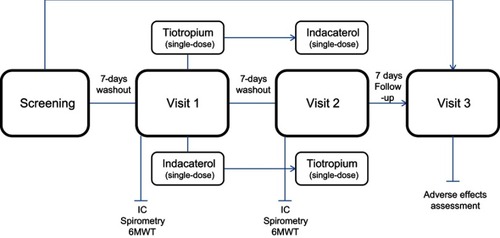
Table 1 Demographics and lung function characteristics on COPD-BE
Table 2 Measurements over time of FEV1, IC, FVC with IND and TIO
Figure 3 Significant increases over all time periods were observed in IC (p<0.0001), after IND was administered. An increase was observed for IC only at 30 mins and 24 hrs after TIO administration. No significant differences were observed among TIO and IND.
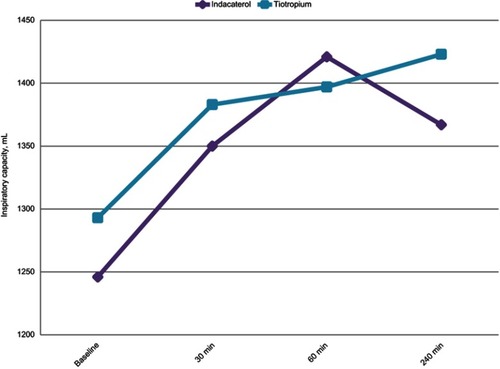
Figure 4 Trough FEV1, FVC and IC after administration a single dose of indacaterol or tiotropium. FEV1 and FVC increased whereas IC decreased with both bronchodilators. p<0.001. However, there were no differences between TIO and IND.
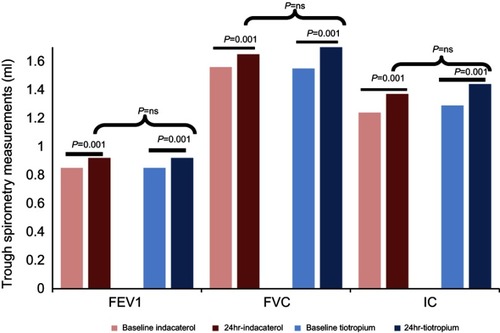
Table 3 Measurements over time of walking distance and dyspnea (end 6MWT) with IND and TIO
Table 4 Subjects achieving minimal clinically important difference (MCID) at different time periods with IND and TIO [n=number of subjects (proportion)]