Figures & data
Figure 1 Flow diagram of study selection process.
Abbreviations: PICOS, population, intervention, comparators, outcomes, and study design; CSRs, clinical study reports; ACCORD, AClidinium in Chronic Obstructive Respiratory Disease COPD; ATTAIN, Aclidinium To Treat Airway obstruction IN COPD patients.
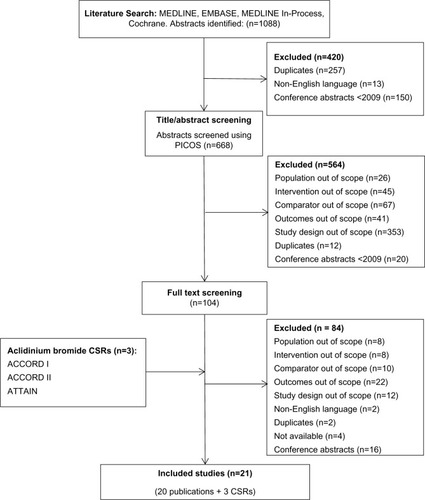
Table 1 Characteristics of included studies
Table 2 Patient characteristics at baseline for the included studies (only arms of interest)
Table 3 Individual studies results included in the network meta-analysis
Figure 2 Network formed by interventions and their direct comparisons included in the analyses.
Abbreviations: GLOW, GLycopyrronium bromide in COPD airways clinical study; OD, once daily; BID, twice a day.
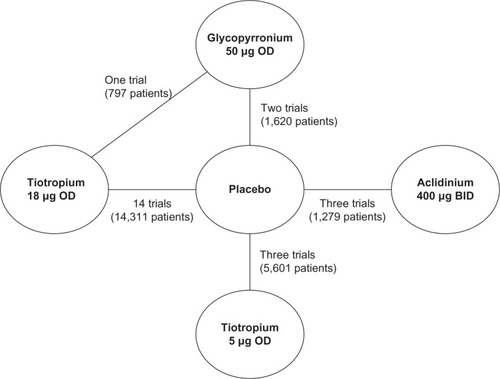
Table 4 Base case: results of the NMA for all treatments versus placebo
Table 5 Results of the network meta-analysis for AB400 versus all other treatments
Figure 3 Forest plot of base case network meta-analysis results for aclidinium.
Abbreviations: AB400, aclidinium 400 μg twice daily; TI05, tiotropium 5 μg bromide once daily; TI018, tiotropium 18 μg bromide once daily; GLYC050, glycopyrronium 50 μg once daily; FEV, forced expiratory volume in 1 second; SGRQ, St George’s Respiratory Questionnaire; TDI, Transition Dyspnea Index.
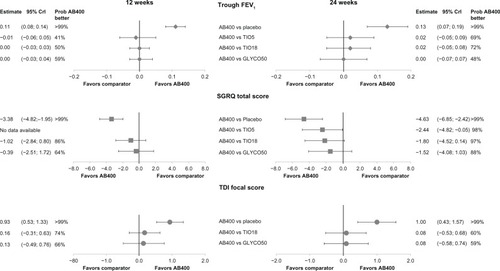
Table S1 Search strategy