Figures & data
Figure 1 Chemical structures of (A) WCK 771 and (B) WCK 2349. Data from De Souza NJ, Gupte SV, Deshpande PK, et al. A chiral benzoquinolizine-2-carboxylic acid arginine salt active against vancomycin-resistant staphylococcus aureus. J Med Chem. 2005;48(16):5232–5242.Citation9
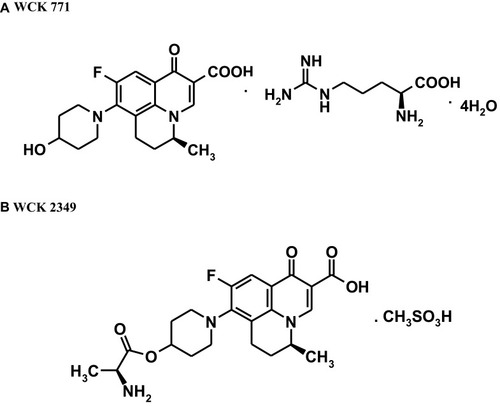
Table 1 In Vitro Activity of WCK 771 Against Staphylococcus aureus (QSSA, QRSA, VISA) Isolates
Table 2 MIC50 and MIC90 of WCK 771 in Comparison to Metronidazole and Clindamycin in Different Anaerobes
Table 3 WCK 771 CLSI QC Ranges for Disk Diffusion and Broth Microdilution Testing
Table 4 In Vivo Efficacy of WCK 771 in a Mouse Systemic Infection Model
Table 5 Mean Levonadifloxacin Cmax and AUC on Day 5 Following Intravenous WCK 771 and Oral WCK 2349 Administered Twice Daily to Healthy Indian Subjects
Table 6 Mean Levonadifloxacin Cmax and AUC on Day 5 Following Intravenous WCK 771 and Oral WCK 2349 Administered Twice Daily to Healthy US Subjects
