Figures & data
Table 1 Patient characteristics
Table 2 Intratumoral therapy experience
Figure 1 X-ray from patient 1.
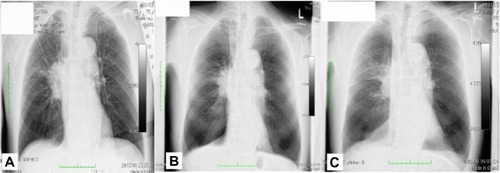
Figure 2 Patient 2 upon diagnosis.
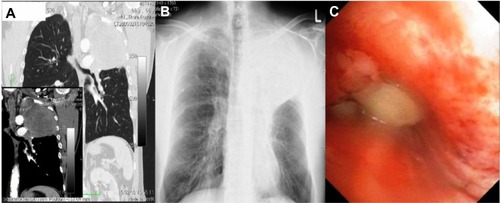
Figure 3 CT images from patient 2.
Abbreviation: CT, computed tomography.
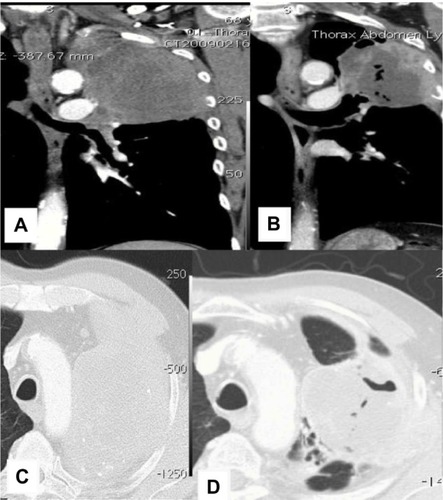
Figure 4 Patient 2 bronchoscopy findings upon diagnosis and after 10 weeks of therapy including four ITC-sessions and three reduced systemic carboplatin doublet applications.
Abbreviation: ITC, intratumoral chemotherapy.
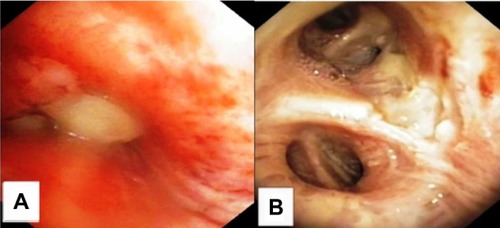
Figure 5 Intratumoral chemotherapy model.
Abbreviation: IF, interstitial fluid.
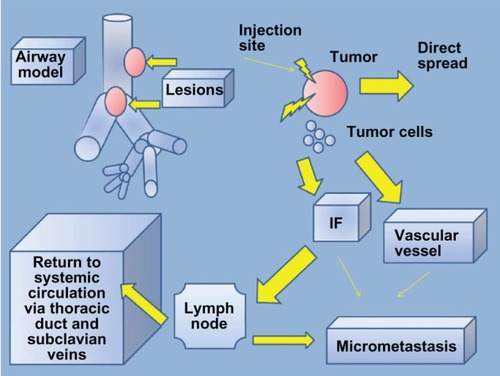
Figure 6 Scheme of proposed intratumoral protocol. Timing of intravenous 4-week-cycle with weekly intratumoral chemotherapy.
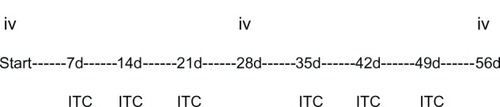
Table 3 Effects and safety features
Table 4 Intratumoral studies using different approaches