Figures & data
Table 2 Taxanes and their combinations recommended by NCCN, ESMO, and AIOM guidelines for triple-negative BCCitation1,Citation16,Citation17
Figure 1 Effect of neoadjuvant chemotherapy in data of triple-negative BCs vs non-triple-negative BCs.Citation23
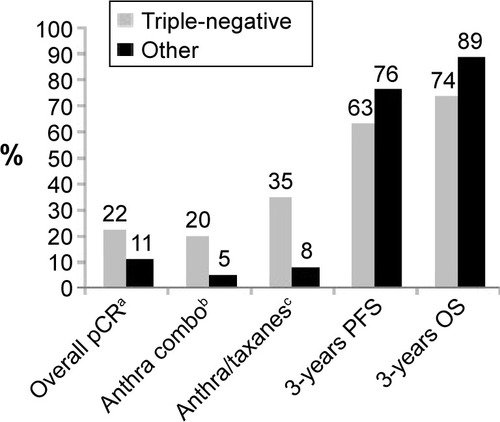
Figure 2 Efficacy parameters in patients with triple-negative BC treated with nab-paclitaxel/bevacizumab/carboplatin.Citation70
Abbreviations: BC, breast cancer; CBR, clinical benefit rate; CI, confidence interval; CR, complete response; ORR, overall response rate; PR, partial response; SD, stable disease.
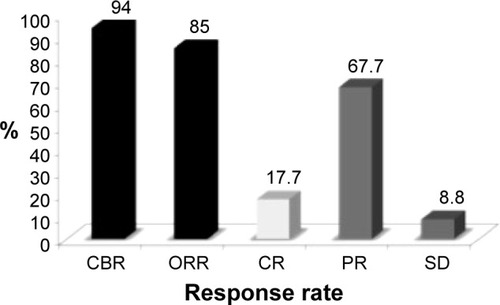
Figure 3 Pathological Response rate in patients treated with conventional paclitaxel or nab-paclitaxel.Citation84
Abbreviation: pCR, pathological complete response.
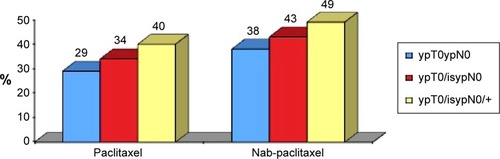
Table 3 Sensorial neuropathy (nab-paclitaxel at 125 mg/m2)Citation84
