Figures & data
Figure 1 Characteristics of DMSA-MNPs (A), anti-CD22-MNPs-DOX (B), and particle size distribution (C) under electron microscopy.
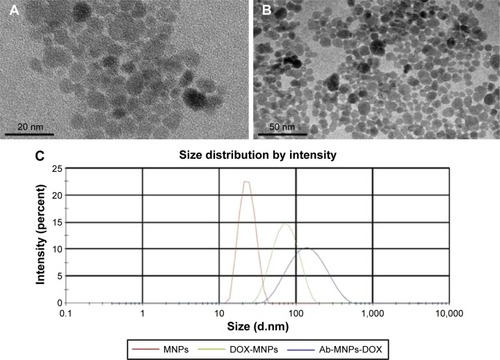
Table 1 Doxorubicin loading in anti-CD22-MNPs-DOX nanoparticles (mean ± standard deviation, n=3)
Figure 2 In vitro release of doxorubicin from anti-CD22-MNPs-DOX.
Abbreviations: DOX, doxorubicin; MNPs, magnetic nanoparticles.
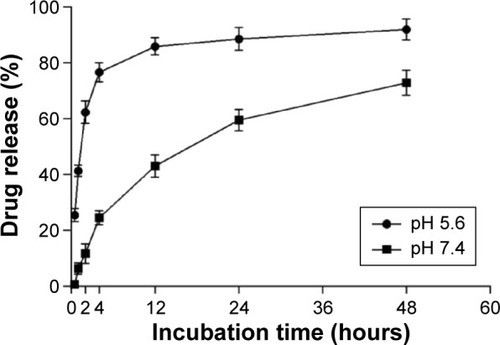
Figure 3 Protein bands of anti-CD22 with and without MNPs-DOX stained by Coomassie Brilliant blue R250.
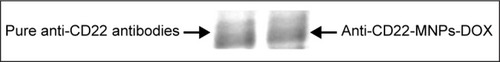
Table 2 IC50 values for DOX-Sol, DOX-MNPs, and anti-CD22-MNPs-DOX at 24 and 48 hours (mean ± standard deviation, n=3)
Figure 4 Cytotoxicity of MNPs (A), doxorubicin, DOX-MNPs, and anti-CD22-MNPs-DOX on Raji cells for 24 hours (B) and 48 hours (C) as determined by Cell Counting Kit-8 assay.
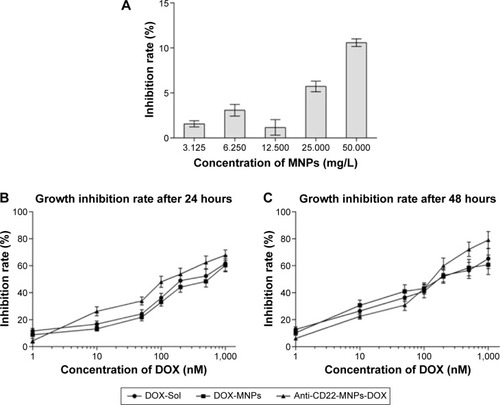
Figure 5 Apoptosis rates of Raji cells for 24 hours by flow cytometry.
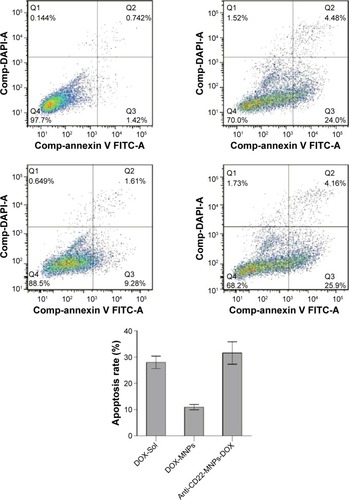
Figure 6 Intracellular accumulation of doxorubicin in Raji cells cultured with the different study formulations for various periods of time. The FI of intracellular doxorubicin (A) was determined by flow cytometry at 488 nm owing to the autofluorescence feature of doxorubicin, and the relative FI is shown in (B).
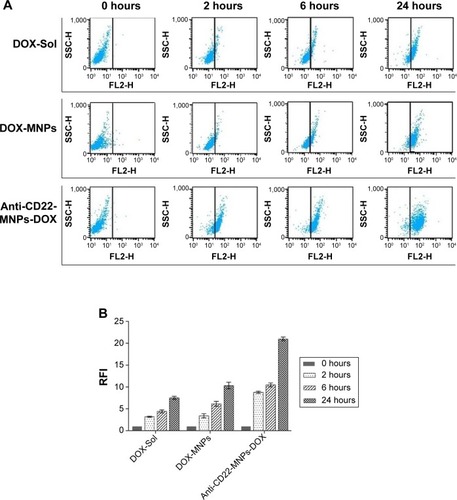
Figure 7 Intracellular accumulation of doxorubicin in Jurkat cells cultured with the different study formulations for 6 hours. X= the average RFI of doxorubicin in Jurkat cells.
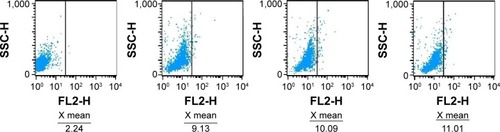
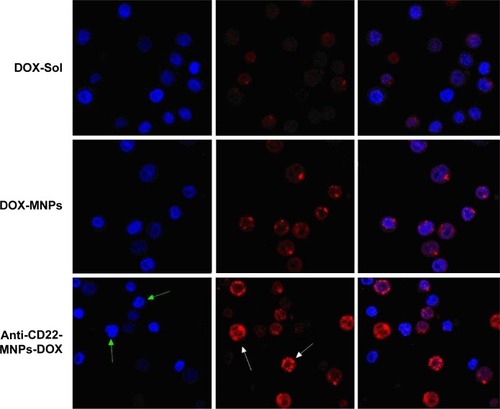
Figure 8 Morphological characterization of Raji cells by laser confocal spectral imaging (200×, DAPI). The signification of arrows shows the increased uptake of anti-cD22-MnPs-DOX in Raji cells. With the green arrows indicating the nuclei with blue fluorescence and white arrows indicate the cytoplasm with red fluorescence.