Figures & data
Figure 1 Overall patient disposition. Analysis of the short-term period included eligible patients who completed three weeks of double-blind treatment with prolonged-release melatonin or placebo. Analysis of the long-term period included patients treated open-label with prolonged-release melatonin for 5 months. Analyses of actigraphic motion recordings included eligible patients who completed 3 weeks of treatment with prolonged-release melatonin and placebo and provided actigraphy data by the end of the treatment period.
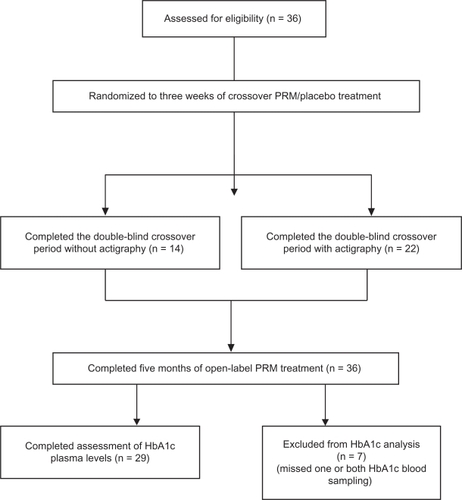
Table 1 Actigraphy-derived sleep parameters (mean ± standard deviation) following 3 weeks of administration of prolonged-release melatonin or placebo (crossover, n = 22)