Figures & data
Figure 1 Schematic illustration of the various CAM structures. Ig-like CAMs are defined by one or more extracellular Ig repeats of 60 to 100 amino acids that form the active adhesion site and a transmembrane segment with a cytoplasmic tail. Classical cadherins have five extracellular cadherin repeats, a single transmembrane domain, and an intracellular tail. Integrin heterodimeric receptors are formed by α and β transmembrane units. Selectins have a short cytoplasmic carboxyl terminus, a transmembrane sequence followed by complement regulatory-like molecules, and an extracellular component containing an epidermal growth factor-like domain and an amino terminal domain that is homologous to C-type lectins. Adapted with permission from Marchetti PJ, O’Connor P. Cellular adhesion molecules in neurology. Can J Neurol Sci. 1997;24(3):201.Citation11
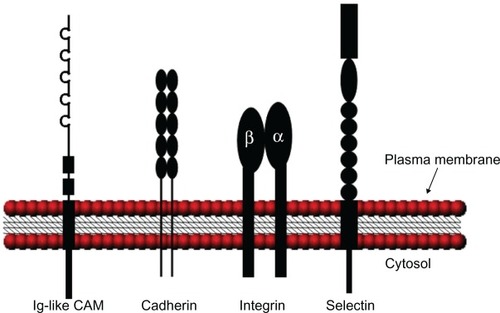
Table 1 Representative CAMs and expression distribution
Table 2 CAMs linked to AD pathological events