Figures & data
Figure 1 Timeline of pivotal Phase III HPV vaccine trials and licensure/registration of the HPV vaccines.
Notes: aTwo-dose schedule separated by six months to adolescents aged <15 years. bData from Iversen et al.Citation57
Abbreviations: 4vHPV, quadrivalent HPV vaccine; 2vHPV, bivalent HPV vaccine; 9vHPV, nonavalent HPV vaccine; FDA, The U.S. Food and Drug Administration; HPV, human papillomavirus; EU, European Union; VLP, virus-like particle.
Figure 2 Countries that have introduced a national human papillomavirus immunization program using any of the three licensed vaccines (91 countries, 47%).
Source: Data from WHO.Citation85
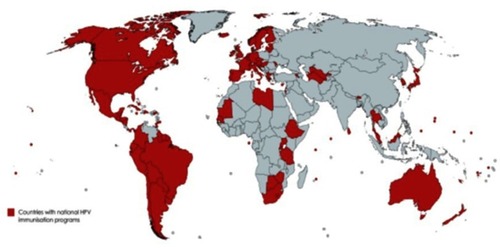
Table 1 Characteristics of HPV VLP vaccines
Table 2 Studies reporting efficacy of 9vHPV against HPV infection and associated diseases
Table 3 Studies reporting immunogenicity and reactogenicity of 9vHPV
