Figures & data
Table 1 Definitions and recommendations for the treatment of Clostridium difficile infection
Table 2 Available antibiotics and investigational new agents for the management of CDI
Figure 1 Rates of clinical cure at end of treatment (primary efficacy endpoint) in the fidaxomicin Phase III trials (studies 003 and 004).Citation22,Citation23
Notes: Modified intent-to-treat: patients underwent randomization and received ≥1 dose of study medication. Per protocol: patients in the modified intent-to-treat population who received ≥3 days of study medication (in cases of failure) or ≥8 days (in cases of clinical cure) with documented adherence to study protocol and who underwent end-of-treatment evaluation.Reproduced with permission from Cornley OA. Current and emerging management options for Clostridium difficile infection: what is the role of fidaxomicin? Clin Microbiol Infect. 2012;18 Suppl 6:28–35Citation72 and Louie TJ, Miller MA, Mullane KM, et al; for OPT-80-003 Clinical Study Group. Fidaxomicin versus vancomycin for Clostridium difficile infection. N Engl J Med. 2011;364(5):422–431.Citation22
Abbreviation: ns, not significant.
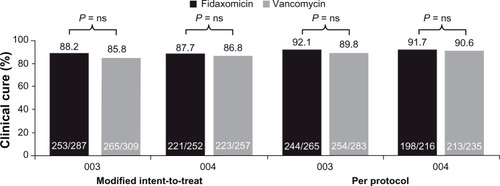
Figure 2 Rates of sustained clinical cure (clinical cure without recurrence of diarrhoea during the 30-day follow-up period) in the fidaxomicin Phase III trials (studies 003 and 004).Citation22,Citation23
Notes: Modified intent-to-treat: patients underwent randomization and received ≥1 dose of study medication. Per protocol: patients in the modified intent-to-treat population who received ≥3 days of study medication (in cases of failure) or ≥8 days (in cases of clinical cure) with documented adherence to study protocol and who underwent end-of-treatment evaluation.Reproduced with permission from Cornley OA. Current and emerging management options for Clostridium difficile infection: what is the role of fidaxomicin? Clin Microbiol Infect. 2012;18 Suppl 6:28–35Citation72 and Louie TJ, Miller MA, Mullane KM, et al; for OPT-80-003 Clinical Study Group. Fidaxomicin versus vancomycin for Clostridium difficile infection. N Engl J Med. 2011;364(5):422–431.Citation22
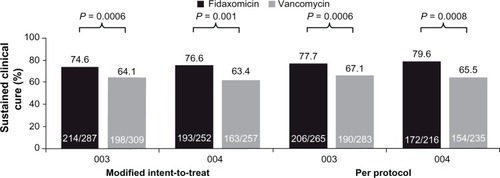
Figure 3 Rates of high-risk patients achieving sustained clinical response (vancomycin versus fidaxomicin).
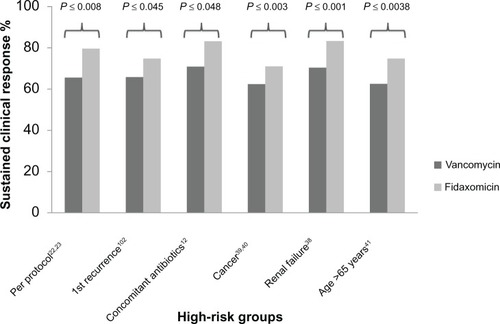
Table 3 Non-antibiotic alternatives and investigational new agents for the management of CDI