Figures & data
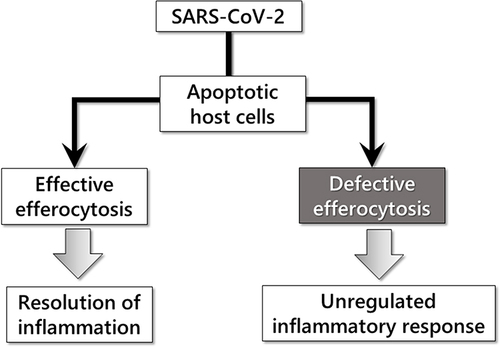
Figure 1 Simplified depiction of the apoptotic/efferocytic process in SARS-COV-2 infection.
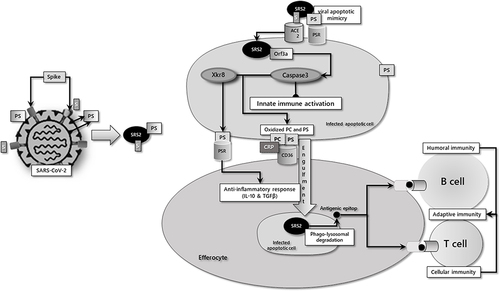
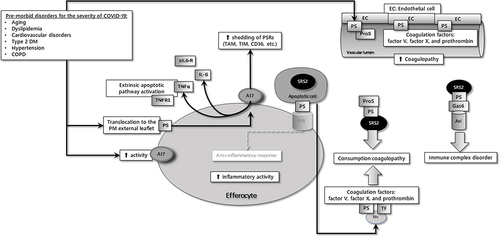
Figure 2 The common characteristics of co-morbid diseases responsible for the severity of COVID-19 are increased PS expression and ADAM-17 (A17) activity in cells.