Figures & data
Table 1 Baseline Characteristics of Patients Treated with PMX-DHP Therapy
Table 2 Results of the Baseline Examinations of Patients Treated with PMX-DHP Therapy (Median)
Figure 2 Time course of the clinical parameters for all patients. Median and interquartile ranges of P/F ratio (A), RR (B), SP-D (C), LDH (D), SOFA (E), CRP (F), IL-6 (G), TARC (H), Neutrocyte (I) and Lymphocyte (J) count. P/F ratio, RR, SP-D, LDH, SOFA, CRP, IL-6, TARC, Neutrocyte and Lymphocyte count were measured before the start of PMX-DHP (day 1), after PMX-DHP was administered 2 times (day 3) and on day 7. Since 2 patients died on day 5, there were no data for 2 cases on day 7. There were no data regarding the SP-D, TARC, and IL-6 for 1 case. CRP, C-reactive protein; IL, interleukin; LDH, lactate dehydrogenase; P/F, PaO2/FIO2; RR, Respiratory ratio; SOFA, sequential organ failure assessment; SP-D, Surfactant Protein-D; TARC, thymus and activation-regulated chemokine; *Kruskal–Wallis test; **Steel-Dwass test.
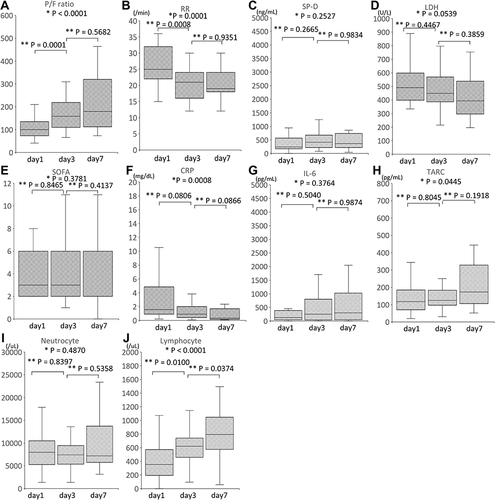
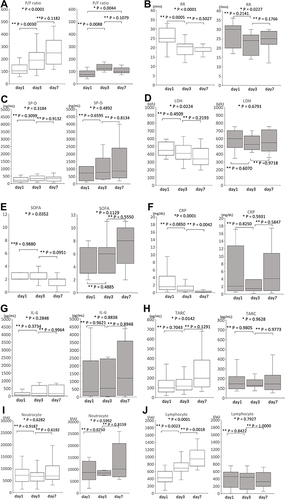
Figure 3 Time course of the clinical parameters for the survival (white) and death (black) groups. Median and interquartile ranges of the P/F ratio (A), RR (B), SP-D (C), LDH (D), SOFA (E), CRP (F), IL-6 (G), TARC (H), Neutrocyte (I) and Lymphocyte (J) count. The P/F ratio, RR, SP-D, LDH, SOFA score, CRP, IL-6, TARC, and Neutrocyte and Lymphocyte count were measured before the start of PMX-DHP (day 1), after PMX-DHP was administered 2 times (day 3), and on day 7. Because 2 patients died on day 5, there were no data for 2 cases in the death group on day 7. There were no data regarding the SP-D, TARC, and IL-6 for 1 case in the death group. CRP, C-reactive protein; IL, interleukin; LDH, lactate dehydrogenase; P/F, PaO2/FIO2; RR, Respiratory ratio; SOFA, sequential organ failure assessment; SP-D, Surfactant Protein-D; TARC, thymus and activation-regulated chemokine; *Kruskal–Wallis test; **Steel-Dwass test.