Figures & data
Figure 1 Median sputum aztreonam lysinate concentrations following nebulized administration of aztreonam to adult patients with cystic fibrosis.Citation31
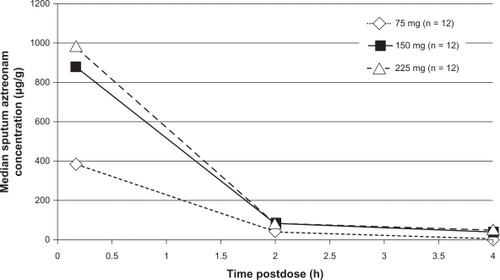
Figure 2 Median sputum aztreonam lysate concentrations following nebulized administration of aztreonam to adolescent patients with cystic fibrosis.Citation31
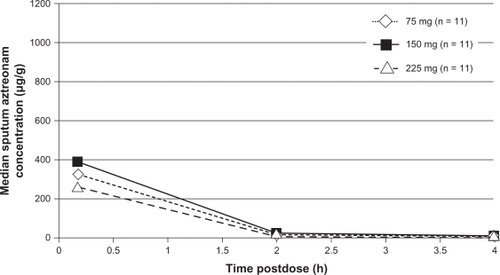
Table 1 Microbiologic endpoints following administration of aztreonam lysine for inhalation to patients with cystic fibrosis for 14 daysCitation32
Table 2 Endpoint results from the Retsch-Bogart et al trialCitation36,Citation37