Figures & data
Figure 1 Schematic strategy of effective designed nanoparticles for advanced stage melanoma. Reprinted from The Lancet Oncology, 15/1, Bombelli FB, Webster CA, Moncrieff M, Sherwood V. The scope of nanoparticle therapies for future metastatic melanoma treatment, e22-e32, Copyright (2014), with permission from Elsevier..Citation110
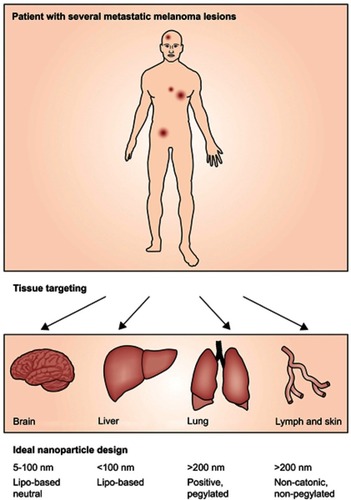
Figure 2 (A) Schematic preparation of CMNPs (B) TEM images of RBC-NPs. The mean particle size (C) and zeta potential (D) of NPs and RBC membranes. (E) SDS-PAGE protein visualization of RBC-membrane, IPH and purified RBC-membrane-camouflaged IPH nanoparticles. Reprinted from Biomaterials, 59, Hao R, Liu J, Li Y, et al.Oxygen self-enriched nanoparticles functionalized with erythrocyte membranes for long circulation and enhanced phototherapy. 59:269, Copyright (2017), with permission from Elsevier.Citation47
Abbreviations: CMNPs, cell membrane-coated nanoparticles; TEM, transmission electron microscopy; RBC-NPs, red blood cell nanoparticles; IPH, human serum albumin nanoparticles.
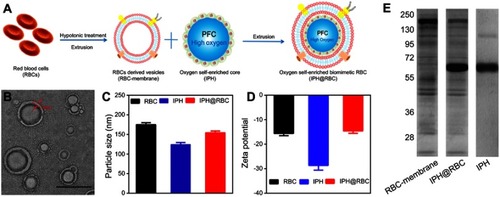
Figure 3 (A) Thermographs of mice injected with the RBC-AuNPs and the pristine PVP-AuNPs after 48 hours. The mice were irradiated with an 850 nm laser (1 W/cm2). (B) Tumor site temperature based on thermographs of mice in (A). Red line indicates the lowest cancerous cells ablation temperature –42°C. (C) Graphs of the average tumor volume. (D) Graphs of the average mouse body weight. (E) Graphs of the mouse survival ratio. (F) Thermographs of tumor-bearing mice under an 808 nm NIR laser exposure (5 W/cm2) for 0–300 seconds. (G) After intravenous injection with MNCs and MNC@RBCs, T2-weighted MR images of tumor-bearing mice were taken at different time points. Blue arrows indicate the tumor locations. Citation41,Citation53 Reprinted from Biomaterials, 92, Ren X, Zheng R, Fang X, et al. Red blood cell membrane camouflaged magnetic nanoclusters for imaging-guided photothermal therapy. 13–24, Copyright (2016), with permission from Elsevier.Citation41 Reprinted with permission from Piao J-G, Wang L, Gao F, You Y-Z, Xiong Y, Yang L. Erythrocyte membrane is an alternative coating to polyethylene glycol for prolonging the circulation lifetime of gold nanocages for photothermal therapy. ACS Nano. 2014;8(10):10414–10425. doi:10.1021/nn503779d. Copyright 2014 American Chemical Society.Citation53
Notes: Fig 3 parts F, G is taken from reference 41, parts A-E is taken from reference 53.
Abbreviations: MNCs, iron oxide magnetic nanoclusters; MNC@RBCs, RBC membrane camouflaged iron oxide magnetic clusters; MF, magnetic field.
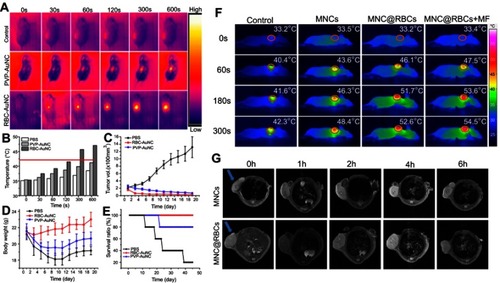
Figure 4 (A) Schematic fabrication process depicting RBC membrane camouflaged programmed co-delivery system. (B) In vivo images of tumor targeting abilities of CNP:Fe, H-CNP:Fe, and RH-CNP:Fe with or without an external magnetic field. (C) Ex vivo optical images of tumors and major organs of tumor-bearing mice sacrificed at 48 hours. (D) Tissue distribution of different NPs in tumor-bearing mice 48 hours post-treatment. (E) The graph of the comparison of signal-over-noise ratios of fluorescence in tumor at different time point. (F) In vivo fluorescence imaging of tumor-bearing nude mice at 2, 24, and 48 hours post-intravenous injection of RBC-PTX (i), anti-EGFR-iRGD and RBC-PTX co-administration (ii) and PRP (iii). The white colored dotted circles indicated the tumor sites. (G) Ex vivo fluorescence imaging of the excised tumors and major organs at 48 hours post-injection. (H) Ex vivo fluorescence imaging of the excised tumors and major organs at 48 hours post-injection of group i and ii. Republished with permission of Royal Society of Chemistry, from Programmed co-delivery of paclitaxel and doxorubicin boosted by camouflaging with erythrocyte membrane, Fu Q, Lv P, Chen Z, et al, 7, 2015; permission conveyed through Copyright Clearance Center, Inc.Citation35 Copyright ©2018. Dove Medical Press. Reproduced from Chen H, Sha H, Zhang L, et al.Lipid insertion enables targeted functionalization of paclitaxel-loaded erythrocyte membrane nanosystem by tumor-penetrating bispecific recombinant protein. Int J Nanomedicine. 2018;13:5347–5359. doi:10.2147/IJN.S165109.Citation56
Notes: Fig 4 parts A-E is taken from reference 35, parts F-H is taken from reference 56.
Abbreviations: PTX, paclitaxel; RGD, Arg-Gly-Asp; DSPE-PEG, 1,2distearoyl-sn-glycero-3-phosphoethanolamine-polyethylene glycol; CNPs, O-carboxymethyl-chitosan (CMC) nanoparticles; PRP, anti-EGFR-iRGD-RBCm-PTX.
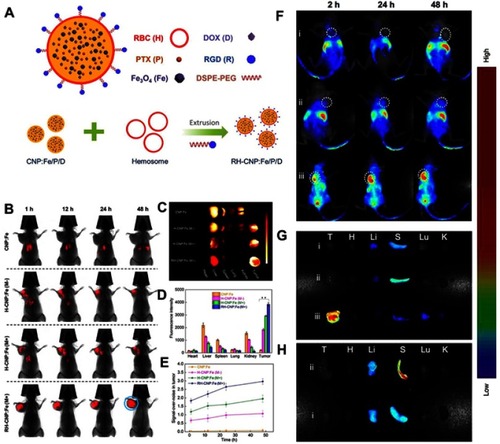
Figure 5 (A, B) TEM images of AuNPs, and macrophage cell membrane-camouflaged AuNPs, respectively. (C) Body weight and (D) tumor volume data were recorded in the course of 3-week treatment. (E) IVIS images of mice injected with cskc-PPiP and cskc-PPiP@ Ma loaded with near-infrared probe. (F) 48 hours post-treatment, 3D reconstruction of fluorescence signal were conducted in cskc-PPiP@Ma-treated mouse. (G) Major organs like heart (H), liver (Li), spleen (S), lung (Lu), kidney (K), and tumor (T) excised from the cskc-PPiP and cskc-PPiP@ Ma injected mouse. (H) Quantification of PTX concentration in organs and tumor tissue excised from mice treated with Taxol, cskc-PPiP/PTX, PPC8/PTX@Ma, and cskc-PPiP/PTX@Ma (n=4). (I) Photographs of the tumor region were taken after 5 minutes of 1 W/cm2 NIR irradiation. (J) Photographs of relevant tumors in accordance with each group in (I). All scale bars are 2 cm. Reprinted with permission from Xuan M, Shao J, Dai L, Li J, He Q. Macrophage cell membrane camouflged Au nanoshells for in vivo prolonged circulation life and enhanced cancer photothermal therapy. ACS Appl Mater Interfaces. 2016;8(15):9610–9618. doi:10.1021/acsami.6b00853. doi:10.1021/nn503779d. Copyright 2016 American Chemical Society.Citation67 Reproduced with permission from Refs.Citation67 Reprinted with permission from Zhang Y, Cai K, Li C, et al. Macrophage-membrane-coated nanoparticles for tumor-targeted chemotherapy. Nano Lett. 2018;18(3):1908–1915. doi:10.1021/acs.nanolett.7b05263. Copyright 2018 American Chemical Society.Citation68
Notes: Fig 5 parts A, B, I, J is taken from reference 67, parts C-H is taken from reference 68.
Abbreviations: MPCM, macrophage cell membrane; AuNSs, gold nanoshells; PPiP, cationic 2aminoethyldiisopropyl group; PPC8, pH-insensitive polymer with neutral octyl group side chains.
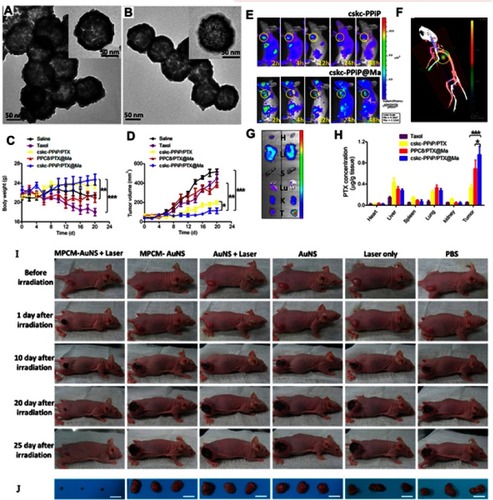
Figure 6 (A) TEM images of (a) bare PLGA NPs, (b) NMVs, and (c) NM-NPs. Scale bar is 100 nm. (B) SDS-PAGE protein tracking of the neutrophils membrane, NM-NPs, and neutrophils, respectively. (C) Western-blot identification of membrane-associated adhesive proteins with and without LPS stimulation. (D) Confocal microscopy analysis of NPs targeting pre-metastatic region in a mouse model (n=3). The injected NPs and NM-NPs were labeled with coumarin-6 (coumarin-6 dosage of 1 mg/kg). Scale bar, 50 μm. (E) H&E staining pictures of lung tissues after mice were treated with saline, free CFZ, NP-CFZ, and NM-NP-CFZ, respectively. Arrows points out metastatic nodules. Scale bar, 5 mm. (F) Histological analysis of cancer cells infiltration in lymph node after the same treatment in (E), n=3. Scale bar, 100 μm. (G) The evaluation of the ability of NM-NP-CFZ, NP-CFZ, and free CFZ to inhibit neutrophils recruitment in mice lungs by immunological staining with S100A9 (brown), n=3. The lung tissues are in accordance with (E). Arrows were pointed to S100A9-positive cells. S100A9 is a cytoplasm protein secreted by neutrophils at the site of inflammation. Scale bar, 100 μm. Reprinted with permission from Kang T, Zhu Q, Wei D, et al.Nanoparticles coated with neutrophil membranes can effectively treat cancer metastasis. ACS Nano. 2017;11:1397–1411. doi:10.1021/acsnano.6b06477. Copyright 2017 American Chemical Society.Citation36
Abbreviations: NMV, Neutrophil-derived membrane vesicles; NM-NP, neutrophil-mimicking nanoparticle; CFZ, carfilzomib.
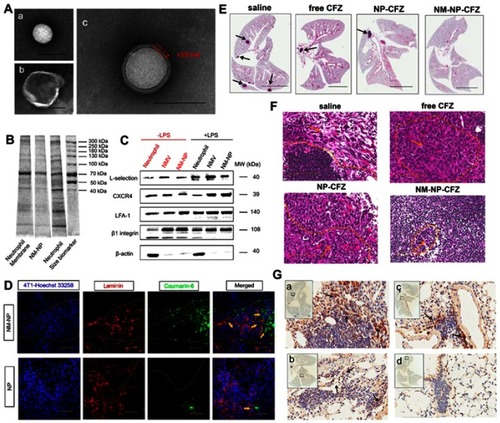
Figure 7 (A) TEM images of ICNPs. (B) Fluorescence images of MCF-7 cells stained with calcein-AM/PI, which were treated with laser, ICG + laser, INPs + laser, or ICNPs + laser. Scale bar, 100 µm. (C) Confocal microscopy images of MCF-7 cells treated with free ICG, INPs, or ICNPs. (D) The infrared thermographic images of ICNPs, INPs, free ICG and PBS after 8 mins continuous laser irradiation at power density 1 W/cm2. (E) Immunogold TEM images of B16-F10, RBC, RBC-B16 membrane, and CuS@[RBC-B16] NPs samples probed for CD47 (red arrows, large gold) and gp100 (yellow arrows, small gold), followed by negative staining with uranyl acetate (scale bars =100 nm). (F) The cell viabilities of B16-F10 cells with different combination of CuS@[RBC-B16] NPs, DOX, NIR. (G) Confocal fluorescent microscopy images of live/dead staining of B16-F10 cells treated with different materials with or without 1,064 nm NIR laser irradiation at power dense 1.0 W/cm2. Scale bar is 100 µm. Reprinted with permission from Chen Z, Zhao P, Luo Z, et al.Cancer cell membrane-biomimetic nanoparticles for homologous-targeting dual-modal imaging and photothermal therapy. ACS Nano. 2016;10(11):10049. doi:10.1021/acsnano.6b04695. Copyright 2016 American Chemical Society.Citation28 Reprinted with permission from Wang D, Dong H, Li M, et al. Erythrocyte-cancer hybrid membrane camouflaged hollow copper sulfide nanoparticles for prolonged circulation life and homotypic-targeting photothermal/chemotherapy of melanoma. ACS Nano. 2018.12(6);5241–5252. Copyright 2018 American Chemical Society.Citation75
Notes: Fig 7 parts A-D, is taken from reference 28, 7 parts E-G is taken from reference 75.
Abbreviations: INPs, PEGylated phospholipid and soybean lecithin shell coated ICG-PLGA core.
![Figure 7 (A) TEM images of ICNPs. (B) Fluorescence images of MCF-7 cells stained with calcein-AM/PI, which were treated with laser, ICG + laser, INPs + laser, or ICNPs + laser. Scale bar, 100 µm. (C) Confocal microscopy images of MCF-7 cells treated with free ICG, INPs, or ICNPs. (D) The infrared thermographic images of ICNPs, INPs, free ICG and PBS after 8 mins continuous laser irradiation at power density 1 W/cm2. (E) Immunogold TEM images of B16-F10, RBC, RBC-B16 membrane, and CuS@[RBC-B16] NPs samples probed for CD47 (red arrows, large gold) and gp100 (yellow arrows, small gold), followed by negative staining with uranyl acetate (scale bars =100 nm). (F) The cell viabilities of B16-F10 cells with different combination of CuS@[RBC-B16] NPs, DOX, NIR. (G) Confocal fluorescent microscopy images of live/dead staining of B16-F10 cells treated with different materials with or without 1,064 nm NIR laser irradiation at power dense 1.0 W/cm2. Scale bar is 100 µm. Reprinted with permission from Chen Z, Zhao P, Luo Z, et al.Cancer cell membrane-biomimetic nanoparticles for homologous-targeting dual-modal imaging and photothermal therapy. ACS Nano. 2016;10(11):10049. doi:10.1021/acsnano.6b04695. Copyright 2016 American Chemical Society.Citation28 Reprinted with permission from Wang D, Dong H, Li M, et al. Erythrocyte-cancer hybrid membrane camouflaged hollow copper sulfide nanoparticles for prolonged circulation life and homotypic-targeting photothermal/chemotherapy of melanoma. ACS Nano. 2018.12(6);5241–5252. Copyright 2018 American Chemical Society.Citation75Notes: Fig 7 parts A-D, is taken from reference 28, 7 parts E-G is taken from reference 75.Abbreviations: INPs, PEGylated phospholipid and soybean lecithin shell coated ICG-PLGA core.](/cms/asset/b47f411a-148f-43c4-ba29-56d7d9c9c9f6/dijn_a_12190849_f0007_c.jpg)
Figure 8 (A) Representative TEM image of negatively uranyl acetate-stained PLT-MNs.Citation79 (B) Representative H&E staining of tumor tissues in tumor-bearing mice with different treatments. Scale bar =50 μm. (C) Images of Ki67 immunohistochemical staining in tumor tissues harvested from the mice in accordance with (B). Scale bar =50 μm. (D) TUNEL staining images of tumor sections from tumor-bearing mice after different treatments. The color blue is nuclei stained by DAPI. Scale bar =20 μm. (E) Representative in vivo IR thermal images of tumor-bearing mice before and after different combinations of treatments. (F) Representative TEM images of RBCNPs, [RBC-P]NPs, and PNPs, negatively stained with vanadium (scale bar =100 nm). (G) Immunogold TEM images of RBCNP, [RBC-P]NP, and PNP samples probed for CD235a (red arrows, large gold) and CD61 (orange arrows, small gold), followed by negative staining with vanadium (scale bars =50 nm). Copyright ©2017. John Wiley and Sons. Reproduced from Rao L, Bu -L-L, Meng Q-F, et al.Antitumor platelet-mimicking magnetic nanoparticles. Adv Funct Mater. 2017;27:9.79.Citation79 Reprinted from Acta Biomaterialia, 80, Zuo H, Tao J, Shi H, He J, Zhou Z, Zhang C. Platelet-mimicking nanoparticles co-loaded with W18O49 and metformin alleviate tumor hypoxia for enhanced photodynamic therapy and photothermal therapy. 296–307, Copyright (2018), with permission from Elsevier.Citation80 Copyright ©2017. John Wiley and Sons. Reproduced from Dehaini D, Wei X, Fang RH, et al.Erythrocyte-platelet hybrid membrane coating for enhanced nanoparticle functionalization. Adv Mater. 2017;29(16):1606209. doi:10.1002/adma.201700681.Citation82
Notes: Fig 8 parts A, E is taken from reference 79, parts B-D is taken from reference 80, parts F,G is taken from reference 82.
Abbreviations: PLT-MNs, PLT membrane-coated particles; [RBC-P]NPs, RBC–platelet hybrid membrane-coated nanoparticles; PNPs, platelet membrane-coated nanoparticles.
![Figure 8 (A) Representative TEM image of negatively uranyl acetate-stained PLT-MNs.Citation79 (B) Representative H&E staining of tumor tissues in tumor-bearing mice with different treatments. Scale bar =50 μm. (C) Images of Ki67 immunohistochemical staining in tumor tissues harvested from the mice in accordance with (B). Scale bar =50 μm. (D) TUNEL staining images of tumor sections from tumor-bearing mice after different treatments. The color blue is nuclei stained by DAPI. Scale bar =20 μm. (E) Representative in vivo IR thermal images of tumor-bearing mice before and after different combinations of treatments. (F) Representative TEM images of RBCNPs, [RBC-P]NPs, and PNPs, negatively stained with vanadium (scale bar =100 nm). (G) Immunogold TEM images of RBCNP, [RBC-P]NP, and PNP samples probed for CD235a (red arrows, large gold) and CD61 (orange arrows, small gold), followed by negative staining with vanadium (scale bars =50 nm). Copyright ©2017. John Wiley and Sons. Reproduced from Rao L, Bu -L-L, Meng Q-F, et al.Antitumor platelet-mimicking magnetic nanoparticles. Adv Funct Mater. 2017;27:9.79.Citation79 Reprinted from Acta Biomaterialia, 80, Zuo H, Tao J, Shi H, He J, Zhou Z, Zhang C. Platelet-mimicking nanoparticles co-loaded with W18O49 and metformin alleviate tumor hypoxia for enhanced photodynamic therapy and photothermal therapy. 296–307, Copyright (2018), with permission from Elsevier.Citation80 Copyright ©2017. John Wiley and Sons. Reproduced from Dehaini D, Wei X, Fang RH, et al.Erythrocyte-platelet hybrid membrane coating for enhanced nanoparticle functionalization. Adv Mater. 2017;29(16):1606209. doi:10.1002/adma.201700681.Citation82Notes: Fig 8 parts A, E is taken from reference 79, parts B-D is taken from reference 80, parts F,G is taken from reference 82.Abbreviations: PLT-MNs, PLT membrane-coated particles; [RBC-P]NPs, RBC–platelet hybrid membrane-coated nanoparticles; PNPs, platelet membrane-coated nanoparticles.](/cms/asset/60901400-9966-4ad4-a044-5548adfbc4ef/dijn_a_12190849_f0008_c.jpg)
Table 1 Biomarkers for various cells
