Figures & data
Figure 1 (A) Schematic diagram of the experimental apparatus for solubility measurement of breviscapine in supercritical CO2; (B) Schematic diagram of the apparatus used for ultrasound-assisted solution-enhanced dispersion by supercritical fluids.
Note: (A): 1, CO2 cylinder; 2, High-pressure syringe pump; 3, Temperature sensor; 4, Pressure sensor; 5, Saturation cell; 6, U-sample collection tube; 7, Circulating pump; 8, Thermostat; (B): 1, CO2 cylinder; 2, Refrigerator; 3, High-pressure pump; 4, Stabilization tank; 5, Pressure sensor; 6, Temperature sensor; 7, Ultrasonic rod; 8, Nozzle; 9, View vessel; 10, Filter; 11, Thermostats; 12, High-pressure constant flow pump; 13, Graduated flask; 14, Separator; 15, Wet gas meter.
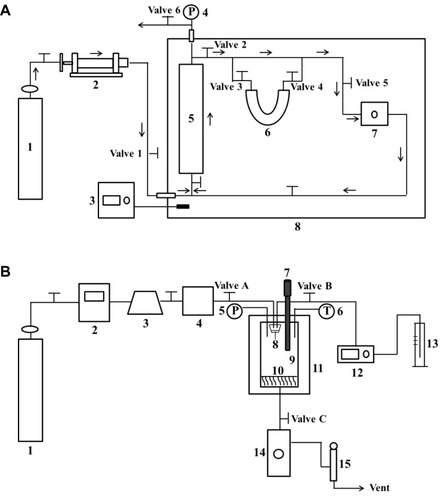
Figure 2 Solubility of breviscapine in supercritical CO2 under various conditions. (A) Pre-saturation time; (B) pressure; and (C) temperature.
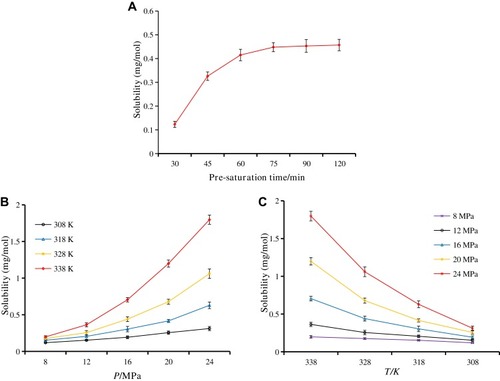
Table 1 Coded Levels and Measured Responses for the 17 Experiment Formulation Runs
Table 2 Comparison of the Experimental and Predicted Values of BRE-MSNs-USEDS Under Predicted Optimum Conditions
Figure 3 Response surface models showing the influence of the factors on the responses.
Notes: Three-dimensional response surface plots showing the effects of (X2) ultrasound power and (X3) value of drug: carrier on the responses of Y1 (A) and Y2 (B). Y1 is the response of the mean particle size. Y3 is the response of drug loading.
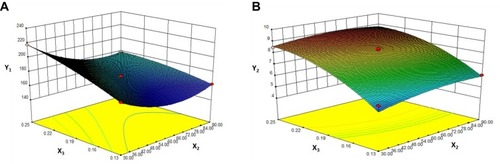
Table 3 The Particle Size, PDI, Zeta Potential and DL of the BRE-MSNs-SIV and BRE-MSNs-USEDS (n=3)
Figure 4 Transmission electron micrograph of BRE-MSNs-SIV and BRE-MSNs-USEDS.
Abbreviations: BRE-MSNs-SIV, breviscapine-loaded mesoporous silica nanoparticles prepared by the solution impregnation-evaporation method; BRE-MSNs-USEDS, breviscapine-loaded mesoporous silica nanoparticles prepared by the ultrasound-assisted solution-enhanced dispersion by supercritical fluids.

Table 4 Material Properties of MSNs Analyzed by Nitrogen Adsorption–Desorption
Figure 5 The nitrogen adsorption–desorption isotherms of the blank MSNs, BRE-MSNs-SIV and BRE-MSNs-USEDS.
Abbreviations: MSNs, mesoporous silica nanoparticles; BRE-MSNs-SIV, breviscapine-loaded mesoporous silica nanoparticles prepared by the solution impregnation-evaporation method; BRE-MSNs-USEDS, breviscapine-loaded mesoporous silica nanoparticles prepared by the ultrasound-assisted solution-enhanced dispersion by supercritical fluids.
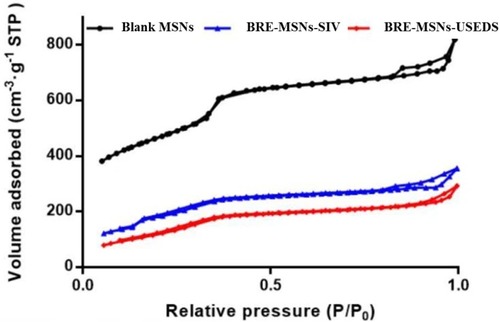
Figure 6 (A) Differential scanning calorimetry analyses and (B) X-ray diffraction analyses of (a) BRE, (b) MSNs, (c) physical mixtures, (d) BRE-MSNs-SIV and (e) BRE-MSNs-USEDS.
Abbreviations: BRE, breviscapine; MSNs, mesoporous silica nanoparticles; BRE-MSNs-SIV, breviscapine-loaded mesoporous silica nanoparticles prepared by the solution impregnation-evaporation method; BRE-MSNs-USEDS, breviscapine-loaded mesoporous silica nanoparticles prepared by the ultrasound-assisted solution-enhanced dispersion by supercritical fluids.
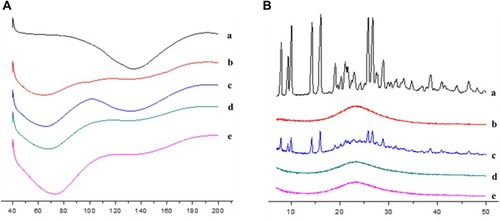
Figure 7 The concentration of residual methanol in BRE-MSNs prepared by different methods.
Abbreviations: BRE-MSNs, breviscapine-loaded mesoporous silica nanoparticles; BRE-MSNs-SIV, breviscapine-loaded mesoporous silica nanoparticles prepared by the solution impregnation-evaporation method; BRE-MSNs-USEDS, breviscapine-loaded mesoporous silica nanoparticles prepared by the ultrasound-assisted solution-enhanced dispersion by supercritical fluids.
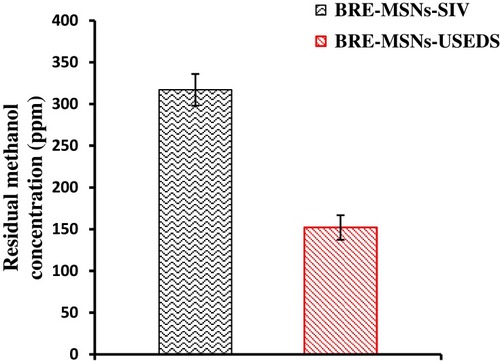
Figure 8 The accelerated stability studies for BRE-MSNs prepared by different methods.
Abbreviations: BRE-MSNs, breviscapine-loaded mesoporous silica nanoparticles; BRE-MSNs-SIV, breviscapine-loaded mesoporous silica nanoparticles prepared by the solution impregnation-evaporation method; BRE-MSNs-USEDS, breviscapine-loaded mesoporous silica nanoparticles prepared by the ultrasound-assisted solution-enhanced dispersion by supercritical fluids.
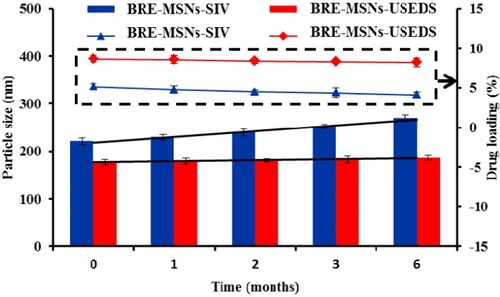
Figure 9 In vitro release of BRE,BRE-MSNs-SIV and BRE-MSNs-USEDS in phosphate buffered solution (pH =6.8).
Abbreviations: BRE, breviscapine; BRE-MSNs-SIV, breviscapine-loaded mesoporous silica nanoparticles prepared by the solution impregnation-evaporation method; BRE-MSNs-USEDS, breviscapine-loaded mesoporous silica nanoparticles prepared by the ultrasound-assisted solution-enhanced dispersion by supercritical fluids.
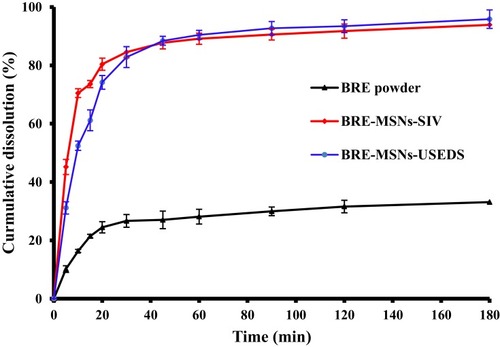
Table 5 Main Pharmacokinetic Parameters of the BRE Powder, BRE-MSNs-SIV and BRE-MSNs-USEDS After Oral Administration to Rats (n = 6)
Figure 10 In vivo plasma concentration-time profiles. Levels of BRE following oral administration of BRE powder suspension, BRE-MSNs-SIV and BRE-MSNs-USEDS (n=6).
Abbreviations: BRE, breviscapine; BRE-MSNs-SIV, breviscapine-loaded mesoporous silica nanoparticles prepared by the solution impregnation-evaporation method; BRE-MSNs-USEDS, breviscapine-loaded mesoporous silica nanoparticles prepared by the ultrasound-assisted solution-enhanced dispersion by supercritical fluids.
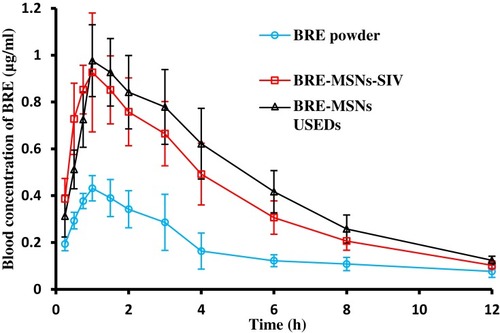
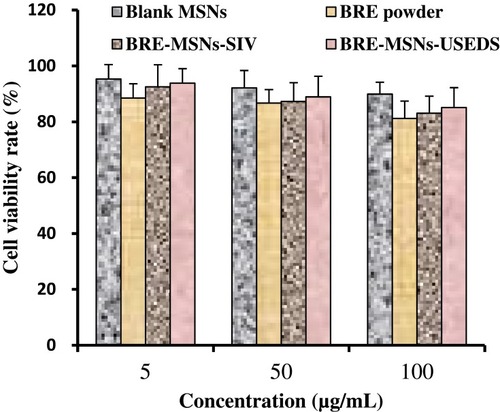
Figure 11 Results of the MTT assay with Caco-2 cells. Data are given as mean ± SD (n=6).
Abbreviations: BRE, breviscapine; MSNs, mesoporous silica nanoparticles; BRE-MSNs-SIV, breviscapine loaded mesoporous silica nanoparticles prepared by the solution impregnation-evaporation method; BRE-MSNs-USEDS, breviscapine loaded mesoporous silica nanoparticles prepared by the ultrasound-assisted solution-enhanced dispersion by supercritical fluids.