Figures & data
Figure 1 Characterization of nanoparticles.
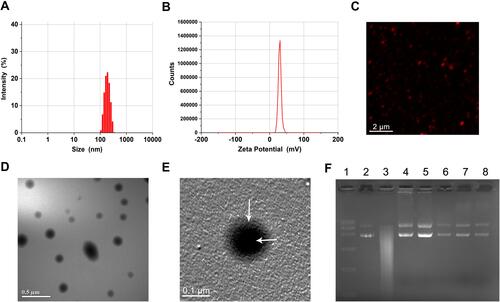
Figure 2 Targeting ability, biosafety, and transfection efficiency of LPHNs-cRGD in vitro.
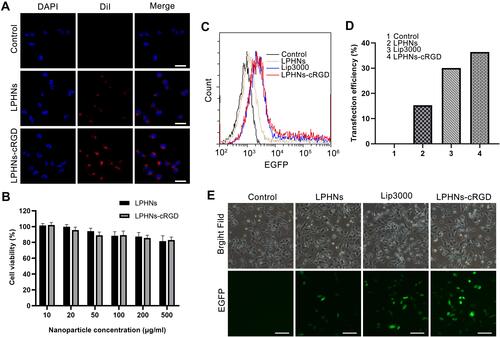
Figure 3 Chemosensitivity testing induced by LPHNspCas9/MGMT-cRGD.
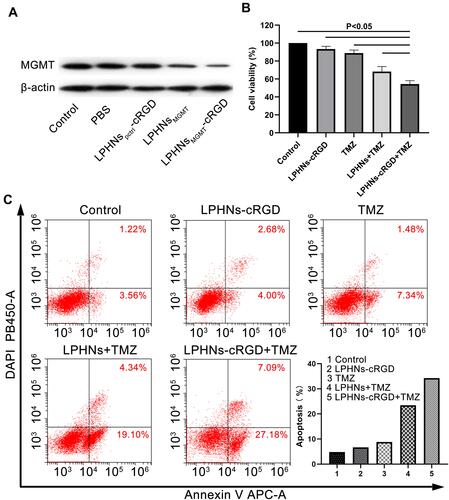
Figure 4 BBB-opening experiment in vivo.
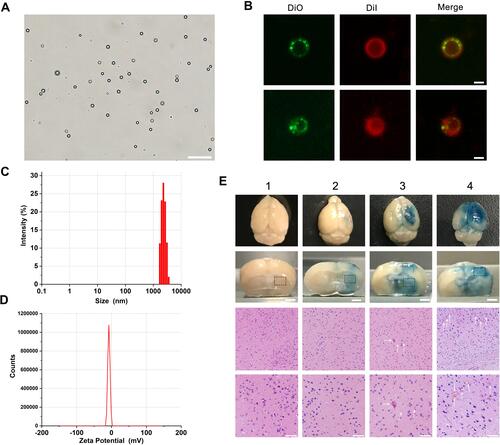
Figure 5 Targeting ability and distribution of MBs-LPHNs-cRGD under FUS irradiation in vivo.
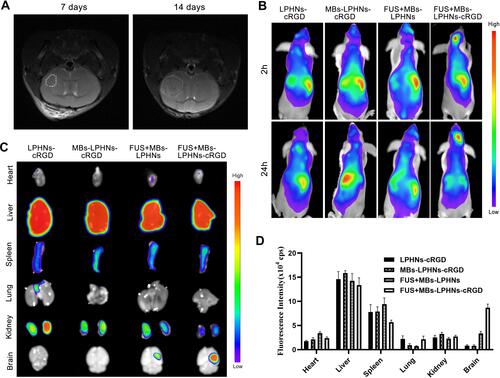
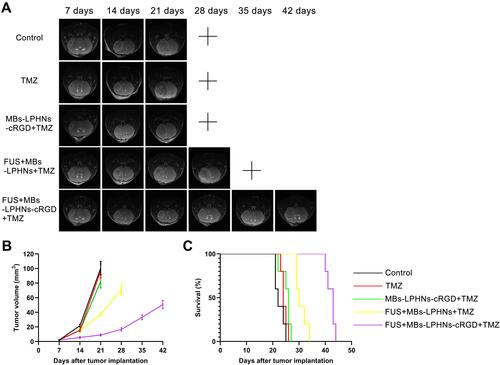
Figure 6 Evaluation of antitumor efficacy in vivo.