Figures & data
Figure 1 The cytotoxicity effects of SPIONs on NSCs. (A) Cell viability measured by the CCK-8 assay. (B) Total GSH content measured by monochlorobimane assay. (C) Activities of glutathione peroxidase and SOD in neural stem cells after 4 hours of exposure. (D) Mitochondrial membrane potential measured in neural stem cells after 4 hours of exposure. *p<0.05. Reproduced with permission from Pongrac IM, Pavičić I, Milić M, et al. Oxidative stress response in neural stem cells exposed to different superparamagnetic iron oxide nanoparticles. Int J Nanomedicine. 2016;11:1701–1715. Copyright 2016, Dove Medical Press.Citation59
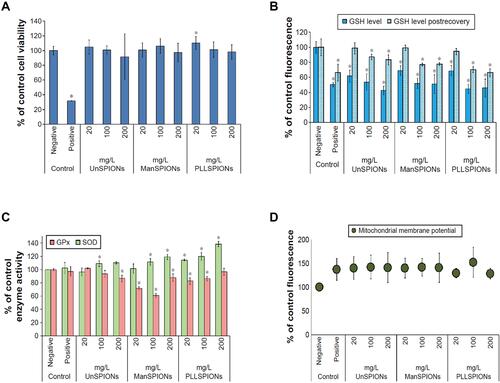
Figure 2 Cellular uptake of silica-coated SPIONs by hAMSCs. (A) The relative amount of non-heme iron inside the cells after incubating with 10, 50 and 100 μg/mL of SPIONs for different time. (B) Immunofluorescence images of untreated cells. (C) Treated with 50 μg/mL of silica-coated SPIONs for 48 h, nanoparticles (green), phalloidin (red) and Hoechst (blue). (D–F) Prussian blue staining of untreated cells (D), SPIONs labeled cells (E and F). Reproduced with permission from Ledda M, Fioretti D, Lolli MG, et al. Biocompatibility assessment of sub-5 nm silica-coated superparamagnetic iron oxide nanoparticles in human stem cells and in mice for potential application in nanomedicine. Nanoscale. 2020;12:1759–1778. Copyright 2020, Royal Society of Chemistry.Citation30
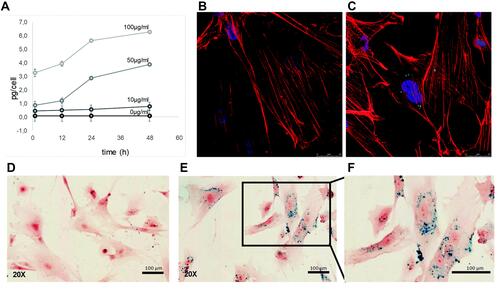
Figure 3 Fluorescence images of Cy5.5 (red)-labeled PCS localized within lysosomes (green) at different time points. (A) Control (a, e, i); PCS exposure for 24 h (b, f, j); PCS exposure for 48 h (c, g, k); PCS exposure for 60 h (d, h, l); (B) Merged fluorescence intensities PCS nanoparticle and lysosome. Reproduced with permission from Lee SH, Park DJ, Yun WS, et al. Endocytic trafficking of polymeric clustered superparamagnetic iron oxide nanoparticles in mesenchymal stem cells. J Control Release. 2020;326:408–418. Copyright 2020, Elsevier.Citation52
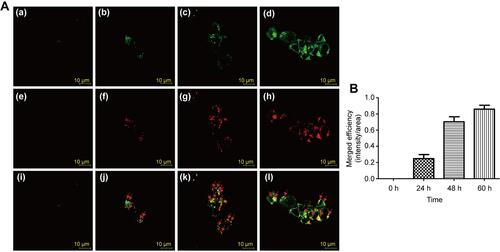
Figure 4 Immunohistology of grafts one-month post-transplantation. (A–D) SPIO-labeled NSCs with nestin (A), β-tubulin (B), MAP2 (C), GFAP (D). (E–H) SPIO-labeled NSCs with nestin (E), β-tubulin (F), MAP2 (G), GFAP (H) staining. Scale Bar=50 mm. Reproduced with permission from Egawa EY, Kitamura N, Nakai R, et al. A DNA hybridization system for labeling of neural stem cells with SPIO nanoparticles for MRI monitoring post-transplantation. Biomaterials. 2015;54:158–167. Copyright 2015, Elsevier.Citation53
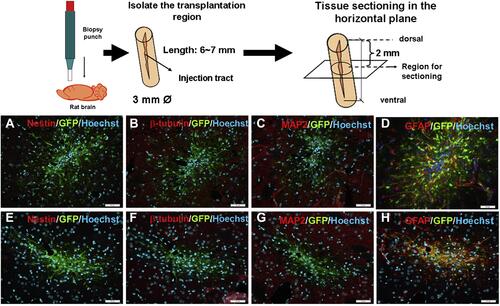
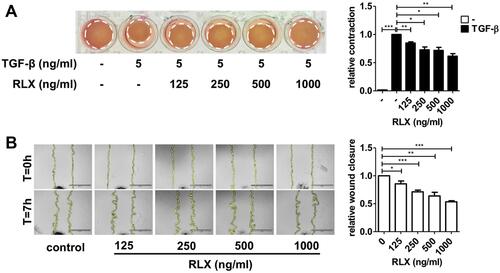
Figure 5 Effect of RLX-SPION on hPSCs contractility and migration. (A) Representative images and quantification. (B) Representative microscopic images and quantification showing. Statistical differences are *p < 0.05, **p < 0.01, ***p < 0.001. Reproduced with permission from Mardhian DF, Storm G, Bansal R, et al. Nano-targeted relaxin impairs fibrosis and tumor growth in pancreatic cancer and improves the efficacy of gemcitabine in vivo. J Control Release. 2018;290:1–10. Copyright 2018, Elsevier.Citation108