Figures & data
Figure 1 Schematic diagram of SpEDS apparatus and brief mechanism for the microencapsulation process.
Abbreviations: HPLC, high-performance liquid chromatography; MTX, methotrexate; SpEDS, suspension-enhanced dispersion by supercritical fluid.
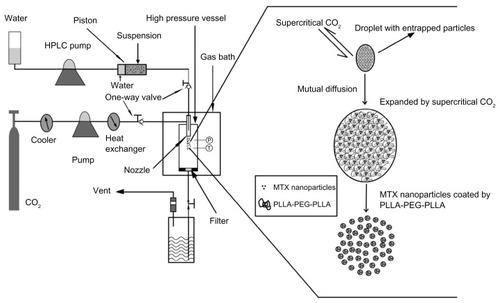
Table 1 Particle size and particle size distribution of (A) methotrexate nanoparticles prepared by SEDS, and MTX-PLLA-PEG-PLLA microspheres from (B) microencapsulation process and (C) coprecipitation process with a drug dose of 10%
Figure 2 Scanning electron micrograph of methotrexate nanoparticles prepared by the solution-enhanced dispersion by supercritical fluid process.

Figure 3 Scanning electron micrograph of MTX-PLLA-PEG-PLLA microspheres from (A) the microencapsulation process and (B) the coprecipitation process.
Abbreviation: MTX-PLLA-PEG-PLLA, methotrexate-poly(L-lactide)-poly(ethylene glycol)-poly(L-lactide).

Figure 4 Fourier transform infrared spectra of methotrexate, PLLA-PEG-PLLA, and MTX-PLLA-PEG-PLLA microspheres from the microencapsulation process and the coprecipitation process.
Abbreviations: MTX, methotrexate; PLLA-PEG-PLLA, poly(L-lactide)-poly(ethylene glycol)-poly(L-lactide).
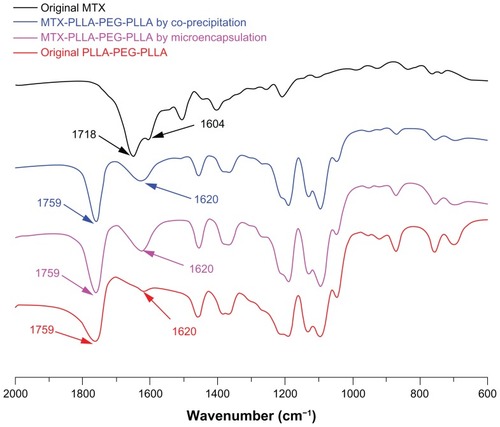
Figure 5 Drug loading and encapsulation efficiency of MTX-PLLA-PEG-PLLA microspheres from the microencapsulation process and the coprecipitation process, with different drug doses.
Abbreviation: MTX-PLLA-PEG-PLLA, methotrexate-poly(L-lactide)-poly(ethylene glycol)-poly(L-lactide).
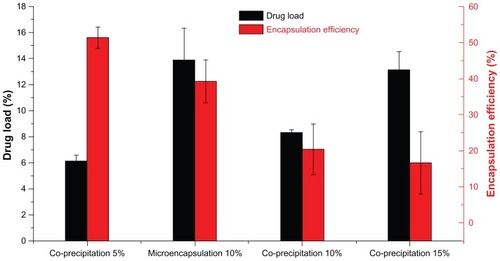
Figure 6 Drug release profiles of MTX-PLLA-PEG-PLLA microspheres from (A) the microencapsulation process and (B) the coprecipitation process with different drug doses.
Abbreviation: MTX-PLLA-PEG-PLLA, methotrexate-poly(L-lactide)-poly(ethylene glycol)-poly(L-lactide).
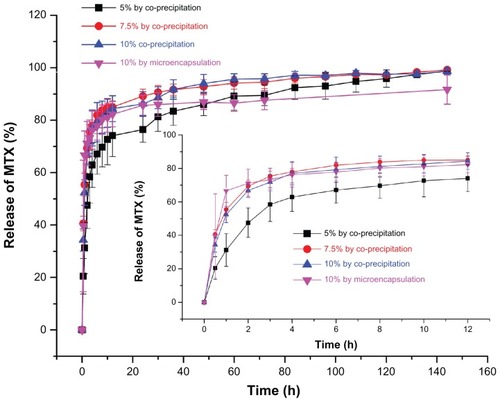
Table 2 Correlation coefficient and standard deviation obtained from the first-order release kinetics fitting of drug release profiles of MTX-PLLA-PEG-PLLA microspheres prepared by the coprecipitation process with different drug doses