Figures & data
Figure 1 Schematic illustrations showing (A) the synthesis of a novel redox-responsive hyperbranched poly(amido amine) (named PCD) and (B) the formation of small interfering RNA (siRNA) ternary complexes with three different mixing orders.
Note: The ternary complexes were prepared in an optimized siRNA/PCD/hyaluronic acid (HA) charge ratio of 1/20/16.
Abbreviations: CBA, N, N′-cystamine bisacrylamide; DMDPTA, N, N′-dimethyldipropylenetriamine.
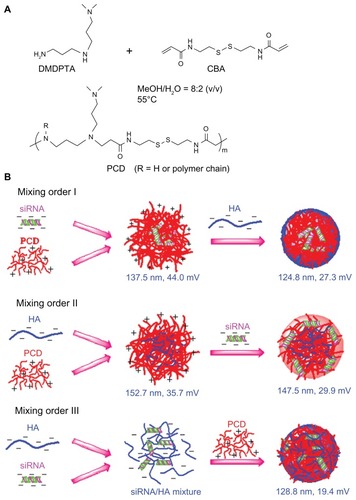
Figure 2 Characterization of the synthesized novel redox-responsive hyperbranched poly(amido amine) (named PCD): (A) proton nuclear magnetic resonance spectrum of PCD, (B) acid-base titration curves of PCD and polyethylenimine (PEI), (C) cytotoxicity of PCD and PEI on MCF-7 human breast cancer cells, and (D) cytotoxicity of PCD and PEI on MDA-MB-231 human breast cancer cells.
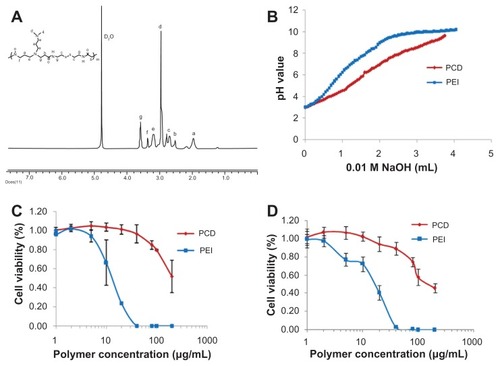
Figure 3 Characterization of small interfering RNA (siRNA) ternary complexes prepared in mixing order I: particle size (A) and zeta potential (B) of siRNA complexes with different amounts of hyaluronic acid (HA) (in charge); (C) gel retardation assay of siRNA/novel redox-responsive hyperbranched poly(amido amine) (named PCD)/HA (C1 and C2) and siRNA/polyethylenimine (PEI)/HA (C3 and C4) ternary complexes with different amounts of HA (from 4 to 20 in charge) in the absence of dithiothreitol (DTT) (C1 and C3) and in the presence of DTT (C2 and C4), the charge ratio of siRNA/polycation is equal to 1/20; (D) cellular uptake of fluorescein-labeled siRNA transfected by various siRNA formulations after incubation at 37°C for 4 hours in MCF-7 and MDA-MB-231 human breast cancer cells.
Note: The untreated cells were used as the control.
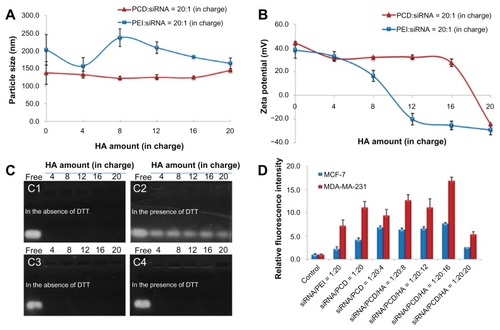
Figure 4 Particle size (A) and zeta potential (B) of small interfering RNA (siRNA) ternary complexes prepared by three mixing orders at different charge ratios of siRNA/novel redox-responsive hyperbranched poly(amido amine) (named PCD)/hyaluronic acid (HA).
Note: *P < 0.01, significant difference between the two groups.
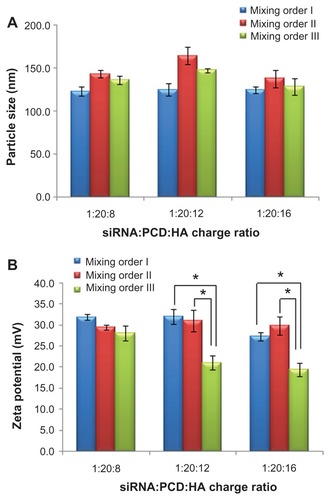
Figure 5 Relative fluorescence intensity of fluorescein-labeled small interfering RNA (siRNA) in various siRNA/novel redox-responsive hyperbranched poly(amido amine) (named PCD)/hyaluronic acid ternary complexes (siRNA/polycation/hyaluronic acid charge ratio of 1/20/16) prepared by different mixing orders.
Note: *P < 0.01, significant difference between the two groups.
Abbreviation: DTT, dithiothreitol.
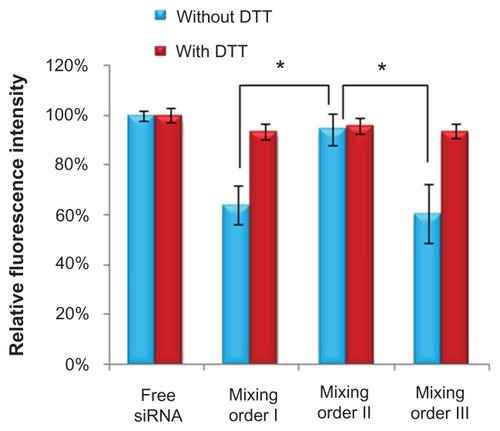
Figure 6 Cellular uptake (4 hours) of fluorescein-labeled small interfering RNA (siRNA) in MCF-7 and MDA-MB-231 human breast cancer cells after incubation with novel redox-responsive hyperbranched poly(amido amine) (named PCD)/siRNA binary complex and various siRNA/PCD/hyaluronic acid ternary complexes (siRNA/polycation/hyaluronic acid charge ratio of 1/20/16) prepared by different mixing orders.
Notes: The untreated cells were used as the control. *P < 0.01, significant difference between the two groups.
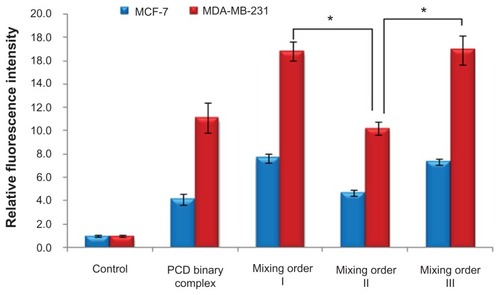
Figure 7 Confocal microscopic images of MDA-MB-231 human breast cancer cells after incubation for 4 hours with (A) fluorescein-labeled small interfering RNA (FAM-siRNA)/polyethylenimine (PEI) binary complex, (B) FAM-siRNA/novel redox-responsive hyperbranched poly(amido amine) (named PCD) binary complex, and FAM-siRNA/PCD/hyaluronic acid ternary complexes prepared by (C) mixing order I, (D) mixing order II, and (E) mixing order III.
Notes: The siRNA/polycation charge ratio is equal to 1/20 for binary complexes, and siRNA/polycation/hyaluronic acid charge ratio is equal to 1/20/16 for ternary complexes. Nuclei and lysosomes were stained with Hoechst 33258 (blue) and LysoTracker® (red), respectively. The scale bars represent 10 μm.
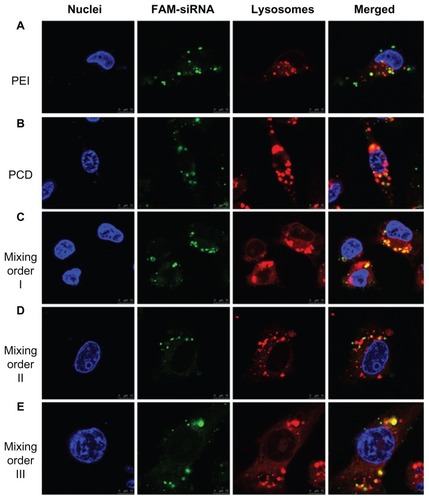
Figure 8 Serum stability of free small interfering RNA (siRNA) and siRNA ternary complexes – siRNA/novel redox-responsive hyperbranched poly(amido amine) (named PCD)/hyaluronic acid charge ratio of 1/20/16 – in the presence of 10% fetal bovine serum: (A) free siRNA, (B) mixing order I, (C) mixing order II, and (D) mixing order III.
Abbreviation: h, hours.
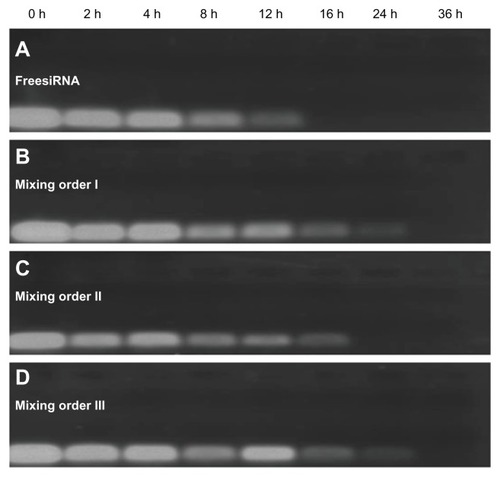
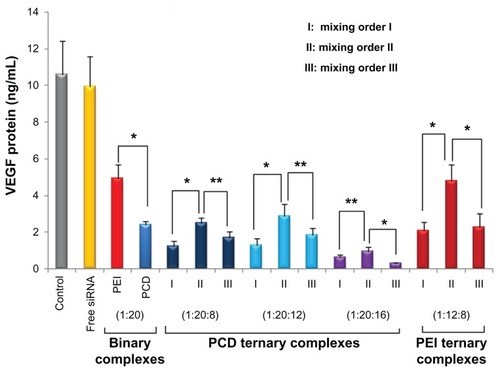
Figure 9 Vascular endothelial growth factor (VEGF) gene silencing activities of various VEGF-directed small interfering RNA (siRNA) complexes in MDA-MB-231 human breast cancer cells. The siRNA ternary complexes were prepared in different charge ratios of siRNA/polycation/hyaluronic acid. For binary complexes, the siRNA/polycation charge ratio is equal to 1/20. The untreated cells were used as the control.
Notes: Results presented as means and standard deviations of three experiments; *P < 0.01, **P < 0.05 between the two groups.
Abbreviations: PCD, novel redox-responsive hyperbranched poly(amido amine); PEI, polyethylenimine.