Figures & data
Table 1 Mean particle size of five kinds of nanoparticles
Figure 1 (A) Drug loading content and (B) encapsulation efficiency of paclitaxel/tetrandrine nanoparticles with different feeding ratios.
Abbreviations: DLC, drug loading content; EE, encapsulation efficiency; Ptx, paclitaxel; Tet, tetrandrine.
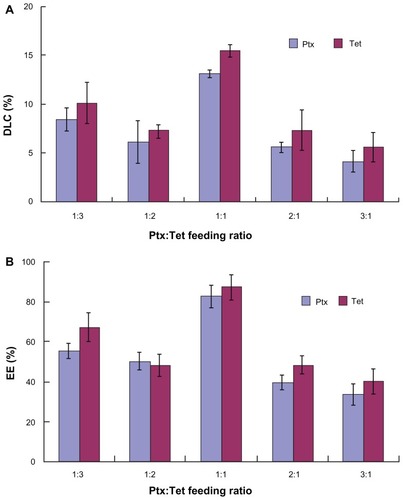
Figure 2 In vitro cytotoxicity of (A) blank nanoparticles and (B) free paclitaxel, paclitaxel plus tetrandrine, and paclitaxel/tetrandrine nanoparticles against H22 cells for 48 hours.
Note: Data are presented as mean ± standard deviation (n = 3).
Abbreviations: Ptx, free paclitaxel; Ptx-Tet, paclitaxel plus tetrandrine; Ptx-Tet np, paclitaxel/tetrandrine nanoparticles.
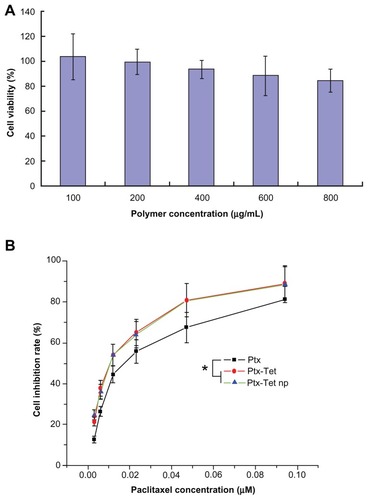
Figure 3 Changes in (A) tumor volume, (B) relative tumor volume, (C) therapeutic group/control group%, and (D) body weight of H22-transplanted imprinting control region mice receiving different therapy treatments. The mice were treated with different protocols on day zero (arrow), ie, when tumor volume measured 100 mm3.
Notes: Data in (A) and (D) are presented as mean ± standard deviation; *P < 0.05 versus the group receiving 10 mg/kg paclitaxel nanoparticles; **P < 0.05 versus the group receiving 5 mg/kg paclitaxel/tetrandrine nanoparticles.
Abbreviations: empty np, empty nanoparticles; P-np 10, paclitaxel nanoparticles in a saline solution at equivalent paclitaxel dose of 10 mg/kg; P/T np, paclitaxel/tetrandrine nanoparticles in a saline solution at equivalent paclitaxel dose of 5, 10, and 20 mg/kg with tetrandrine dose set at 10 mg/kg; Ptx 10, free paclitaxel at a dose of 10 mg/kg; saline, vehicle; T/C, therapeutic group/control group.
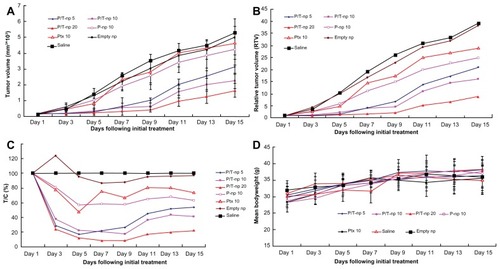
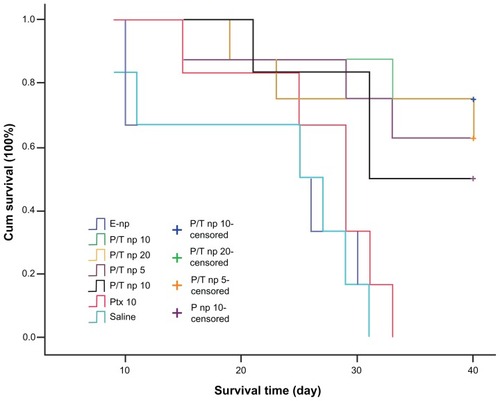
Figure 4 Kaplan–Meier curves showing the survival of tumor-bearing mice treated with different protocols.
Note: The different agents were delivered intratumorally when tumor volume measured 100 mm3.
Abbreviations: cum, cumulative; empty np, empty nanoparticles; P-np 10, paclitaxel nanoparticles in a saline solution at equivalent paclitaxel dose of 10 mg/kg; P/T np, paclitaxel/tetrandrine nanoparticles in a saline solution at equivalent paclitaxel dose of 5, 10, and 20 mg/kg with tetrandrine dose set at 10 mg/kg; Ptx 10, free paclitaxel at a dose of 10 mg/kg; saline, vehicle.