Figures & data
Figure 1 Cortical bone trajectory (CBT) screw placement under the guidance of 3D-printed templates and cement injection.
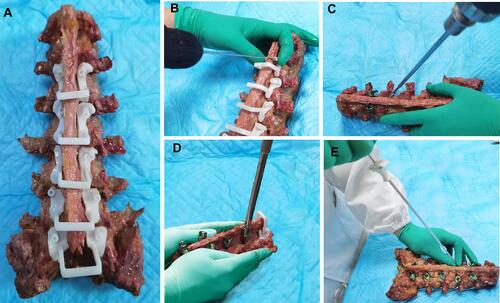
Table 1 Information and Characterization of Cadaver Specimens
Figure 2 Radiological evaluation of cement-augmented cortical bone trajectory (CBT) screws.
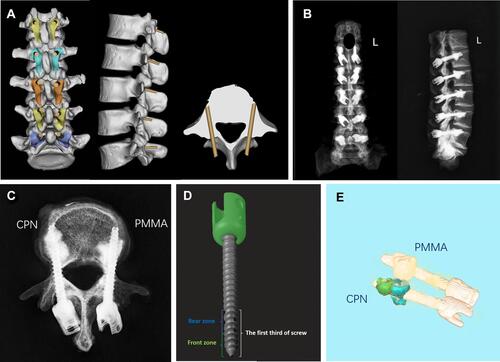
Figure 3 Biomechanical testing of cemented cortical bone trajectory screws.
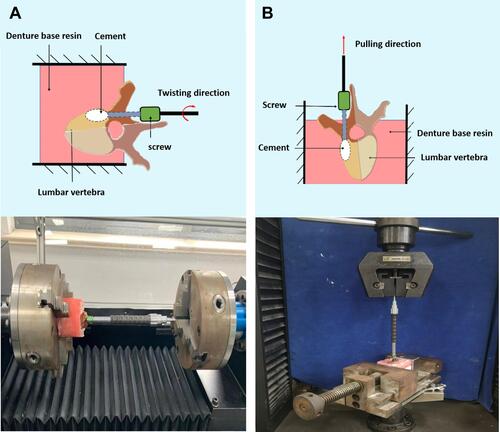
Figure 4 The results of the setting time and rheological properties.
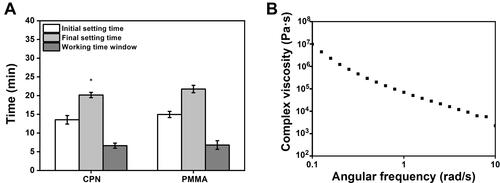
Figure 5 The results of compressive tests.
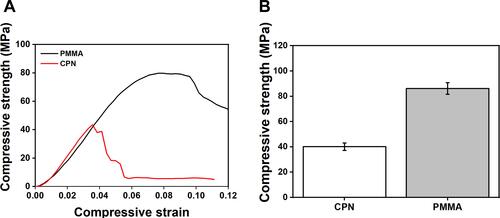
Figure 6 Results of biomechanical tests.
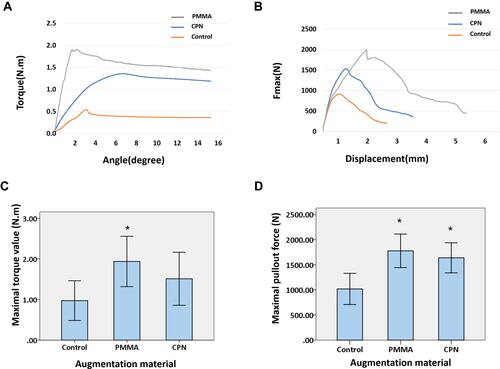
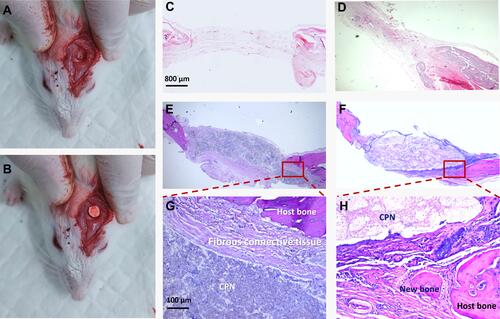
Figure 7 Histological analysis of the calcium phosphate–based nanocomposite (CPN) after implantation in a rat cranial defect model.